Of course. The discovery of the glymphatic system in 2012 was a landmark moment in neuroscience, fundamentally changing our understanding of how the brain maintains its health. Here is a detailed explanation of that discovery.
The Discovery of the Brain’s Hidden Cleaning System
For centuries, the brain was believed to be an immune-privileged organ, isolated from the body’s lymphatic system and lacking a dedicated waste clearance mechanism. It was a mystery how the brain, with its intense metabolic activity, managed to rid itself of toxic proteins and cellular debris. This mystery began to be solved just over a decade ago by a team at the University of Rochester Medical Center, led by Dr. Maiken Nedergaard .
In August 2012, the team published a groundbreaking study in the journal Science Translational Medicine that unveiled a previously unknown system for waste removal in the brain, which they named the “glymphatic system” . The name itself is a clue to its function: “gly” refers to the glial cells (specifically astrocytes) that manage the system, and “lymphatic” denotes its role as a waste-clearance pathway, similar to the lymphatic system in the rest of the body .
The “Aha!” Moment: Seeing the System in Action
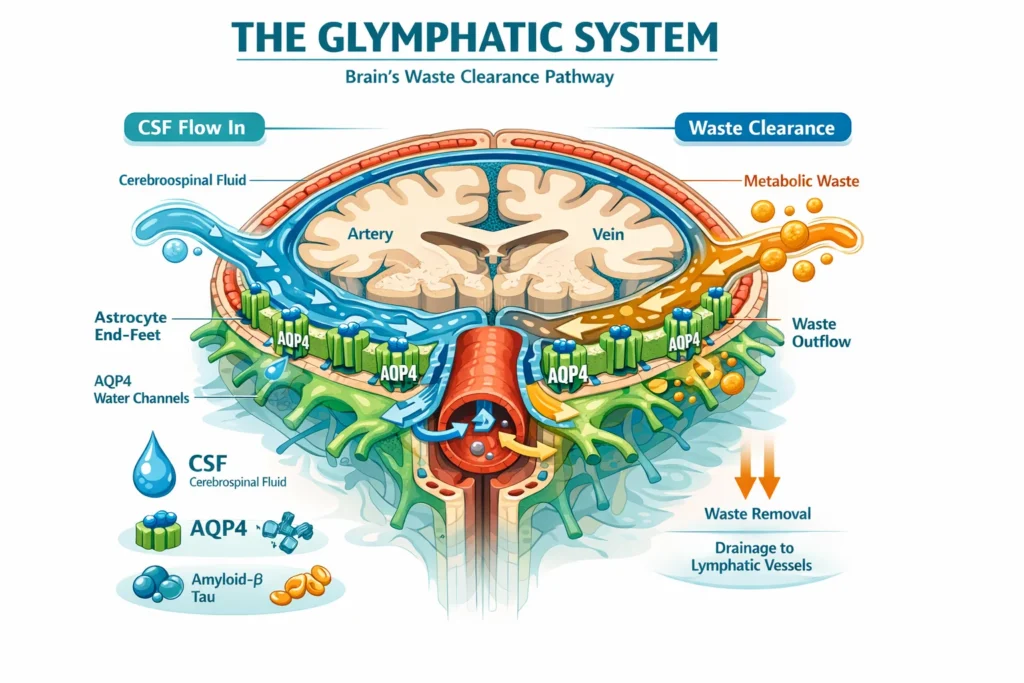
The key to the discovery was technological. Previous attempts to study brain waste clearance were limited because the system collapses and becomes impossible to study once the brain is removed from the skull or after an animal dies . The Rochester team utilized a powerful imaging technique called two-photon microscopy, which allowed them to look into the living brains of mice and observe the flow of cerebrospinal fluid (CSF) in real-time .
What they saw was a highly organized process:
- A Highway System: They discovered that the brain’s blood vessels are not just for blood. They are surrounded by intricate, tunnel-like spaces that form a network of conduits throughout the brain .
- The Gatekeeper: They observed that specialized glial cells called astrocytes use projections known as “end-feet” to line these tunnels. These end-feet are densely packed with water channels called aquaporin-4 (AQP4) , which act as valves to facilitate the rapid flow of CSF through the brain tissue .
- A One-Way Street: The researchers mapped the flow, showing that CSF is pumped into the brain along the spaces surrounding arteries. It then washes through the brain’s interstitial space, collecting metabolic waste. Finally, this now “dirty” fluid is collected along the spaces surrounding veins and flushed out of the brain .
Why the Discovery Was So Significant
The discovery of the glymphatic system was revolutionary for several reasons:
- It Solved a Long-Standing Mystery: It finally explained how the brain, which produces significant amounts of metabolic waste, clears itself of toxins like amyloid-beta, the protein that forms the hallmark plaques in Alzheimer’s disease . The study’s first author, Dr. Jeffrey Iliff, found that more than half of the amyloid-beta cleared from a healthy mouse brain is removed via this newly discovered pathway .
- It Explained the Purpose of Sleep: The research showed that the glymphatic system is primarily active during sleep, suggesting that one of the key reasons we sleep is to allow our brains to be “power-washed” and cleansed of waste that accumulates during waking hours .
- It Opened New Avenues for Treating Neurodegenerative Disease: The discovery provided a new framework for understanding diseases like Alzheimer’s, Parkinson’s, and others. It suggested that these conditions might not just be about the overproduction of toxic proteins, but also about the failure of the brain’s waste clearance system . As Dr. Iliff noted, if the glymphatic system fails, waste may begin to accumulate in the brain, potentially leading to the amyloid deposits seen in Alzheimer’s disease .
The 2012 discovery, initially in mice, has since been explored in other species, including humans, dolphins, and non-human primates, confirming its fundamental importance to central nervous system health . It has transformed our understanding of brain physiology and opened up exciting new possibilities for diagnosing, preventing, and treating a wide range of neurological conditions .
For years, the brain was thought to be an immune-privileged organ, isolated from the body’s typical cleanup crews. However, the discovery of the glymphatic system just over a decade ago revolutionized our understanding of neurological health. This system acts as the brain’s dedicated housekeeper, a network that flushes out metabolic waste and toxins, particularly during deep sleep . Now, researchers are learning to read the signals of this system by analyzing the very fluid that flows through it—the cerebrospinal fluid (CSF). Emerging evidence suggests that specific components in the CSF could serve as early biomarkers of impaired waste clearance, potentially years before the onset of diseases like Alzheimer’s and Parkinson’s .
The Silent Plumber: Why Clearance Efficiency Matters
The glymphatic system works by channeling CSF from the subarachnoid space into the brain along arteries. This fluid then swaps with interstitial fluid (ISF), collecting soluble proteins and metabolites before being drained away along veins . This process is heavily dependent on sleep; the system is most active during these periods, effectively power-washing the neural tissue .
When this system fails, metabolic byproducts accumulate. In Alzheimer’s disease, this means a buildup of amyloid-beta and tau proteins. In Parkinson’s, it involves the aggregation of alpha-synuclein. The “Impaired Clearance Hypothesis” posits that this dysfunction—rather than just overproduction of toxins—is a primary driver of neurodegeneration . If we can measure how well this system is working, we might identify individuals at risk long before cognitive symptoms appear.
Aquaporin-4 Fragments: The Astrocytic Gatekeeper
One of the most promising candidates for a CSF biomarker is Aquaporin-4 (AQP4) . This water channel is densely packed on the end-feet of astrocytes, the cells that line the brain’s blood vessels. AQP4 facilitates the rapid influx of CSF from perivascular spaces into the brain parenchyma, making it a critical valve for the entire clearance system .
Recent studies have shown that AQP4 is not static. In neurodegenerative conditions, it becomes dysregulated. A pivotal 2024 study published in Human Brain Mapping found that CSF levels of AQP4 were significantly higher in patients with Alzheimer’s disease and other neurodegenerative dementias compared to cognitively unimpaired individuals . This increase is thought to reflect a compensatory mechanism or a sign of mislocalization—where AQP4 detaches from the astrocytic end-feet and is shed into the CSF. This makes AQP4 fragments in the CSF a direct indicator of glymphatic system integrity, specifically within the brain parenchyma .
Perivascular Drainage Proteins: Markers of Clogged Highways
The perivascular spaces (PVS) are the highways through which CSF travels. When these spaces become clogged or fibrotic, drainage fails. Research into conditions like Cerebral Amyloid Angiopathy (CAA) and post-stroke recovery has highlighted the role of extracellular matrix proteins in this process.
Following an ischemic stroke, for instance, an abnormal pattern of fibrosis can occur within the vascular basement membrane. This perivascular fibrosis, driven by factors like Transforming Growth Factor-beta (TGF-β), physically impedes the drainage of amyloid-beta along the arteries . While much of this research is still in animal models, the presence of fibrotic markers or extracellular matrix proteins in the CSF could theoretically serve as a biomarker for “clogged” drainage routes. These proteins would indicate not just a cellular problem (like AQP4 dysfunction) but a structural obstruction in the perivascular pathways.
Sleep-Related Metabolic Byproducts: The Nightly Tally
The link between sleep and clearance is so strong that the byproducts of metabolism cleared during sleep can serve as indirect markers of efficiency. While amyloid-beta and tau are the most famous waste products, other metabolites are gaining attention.
For example, disruptions in the flow of CSF are linked to changes in the concentration of metabolites like orexin (a regulator of wakefulness) and various interleukins . Furthermore, in Parkinson’s disease, researchers are investigating CSF flow dynamics directly. A 2026 study in NPJ Parkinson’s Disease used advanced MRI to measure “CSF pseudo-diffusivity” and found that Parkinson’s patients exhibit significantly reduced CSF flow dynamics within the ventricles and sulci . This suggests that in PD, the physical movement of CSF—the “river” that carries waste—is stagnant. While this study used imaging, the stagnation of flow would theoretically correlate with higher concentrations of sleep-related metabolic byproducts that failed to be transported away overnight.
Towards a Clinical Future: The Relevance to Disease
The convergence of these biomarkers points to a paradigm shift in how we view neurodegeneration. Rather than waiting for plaques and tangles to form, we may soon be able to detect the conditions that allow them to form.
- Alzheimer’s Disease: A patient might present with high CSF AQP4 (indicating astrocytic dysregulation) and enlarged perivascular spaces visible on MRI (indicating upstream clogging), signaling a brain that is struggling to clear amyloid, even if amyloid PET scans are currently negative .
- Parkinson’s Disease: A patient with REM sleep disorder (a common precursor to PD) might show reduced CSF flow dynamics or alterations in CSF proteins linked to the locus coeruleus, a brainstem region involved in regulating CSF flow that degenerates in PD .
The study of CSF biomarkers for brain waste clearance is still in its infancy, but it is rapidly maturing. By analyzing fragments of the brain’s plumbing system (AQP4), the structural integrity of its drainage highways (perivascular proteins), and the efficiency of its nightly cleanup (metabolites), we are learning to listen to the brain’s private communications. Ultimately, impaired waste clearance may not just be a consequence of neurodegeneration, but a precipitating event, making these CSF biomarkers our best chance for early detection and intervention .