We spend about a third of our lives asleep, and for a long time, scientists viewed sleep primarily as a period of rest for the body and mind. But groundbreaking research over the past decade has revealed something far more active: while you sleep, your brain engages in a nightly deep-clean process that removes toxic waste built up during the day. This mechanism, known as the glymphatic system, acts like the brain’s own waste disposal unit, and understanding it could hold keys to preventing neurodegenerative diseases like Alzheimer’s.
Here are some fascinating, evidence-based facts about how your brain cleans itself every night through the glymphatic system — the brain’s built-in waste-clearance mechanism. These draw from key discoveries and recent research (up to 2025–2026).
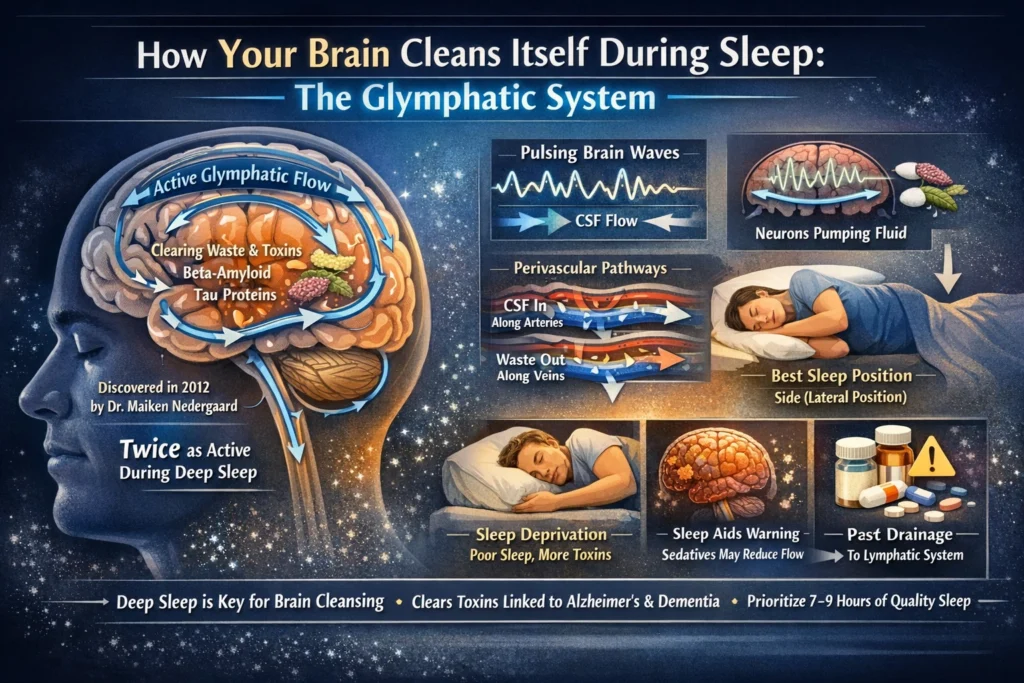
- The glymphatic system was discovered in 2012 by neuroscientist Maiken Nedergaard and her team at the University of Rochester. It functions as the brain’s equivalent of the lymphatic system, using cerebrospinal fluid (CSF) to flush out metabolic waste, toxins, and proteins like beta-amyloid and tau — which build up during wakefulness and are linked to Alzheimer’s and other neurodegenerative diseases.
- This “brain washing” process is dramatically more efficient during sleep, especially deep non-REM sleep (slow-wave sleep or stage N3). Studies show glymphatic clearance can be up to twice as active (or more) during sleep compared to wakefulness, as brain cells shrink slightly — expanding interstitial spaces by up to 60% — allowing better fluid flow.
- Neurons act as miniature pumps during sleep. Recent 2024–2025 research (including from Washington University and Nedergaard’s lab) revealed that synchronized electrical activity — like delta waves in deep sleep and norepinephrine-driven oscillations — generates rhythmic pulses that propel CSF through brain tissue, driving waste removal. In essence, “neurons that fire together shower together.”
- The system relies on perivascular spaces (channels around blood vessels) and aquaporin-4 (AQP4) water channels on astrocytic endfeet. CSF enters along arteries, mixes with interstitial fluid to pick up waste, and exits along veins — ultimately draining to the body’s lymphatic system. Sleep synchronizes this with slow brain waves, reduced norepinephrine initially, and vascular pulsations.
- Poor sleep impairs clearance: Sleep deprivation or disruption reduces glymphatic flow, leading to higher levels of toxic proteins in the brain. Human studies link chronic poor sleep to accelerated buildup of amyloid-beta and tau, increasing risks for dementia, Parkinson’s, and cognitive decline.
- Not all sleep stages are equal — deep slow-wave sleep is prime time for glymphatic activity, boosted by higher delta waves and lower heart rate/beta activity. Some research shows activity in REM and even during wake-up transitions, but non-REM (especially deep) dominates the nightly detox.
- Sleep position matters: Sleeping on your side (lateral position) appears more efficient for glymphatic clearance than prone or supine positions, based on studies in animals and implications for humans.
- Certain sleep aids can interfere: A 2025 study found that zolpidem (Ambien) reduces glymphatic clearance by about 30% in mice by disrupting norepinephrine-mediated vascular oscillations — highlighting how some medications might hamper natural brain cleaning.
- The glymphatic system isn’t just for waste removal — it also delivers nutrients like glucose and lipids to brain cells. When impaired (e.g., by aging, high blood pressure, brain injury, or poor sleep), it contributes to neuroinflammation and disease progression.
- Recent human imaging (2024–2025) — including new monitoring devices with head caps tracking fluid shifts — confirms the system operates in people during sleep, not just in animal models. This supports why prioritizing 7–9 hours of quality sleep (with ample deep stages) is one of the best ways to support long-term brain health.
These facts highlight sleep as far more than rest — it’s your brain’s essential nightly maintenance routine. Prioritizing good sleep hygiene could be a powerful defense against cognitive decline.
Discovery of the Brain’s Hidden Cleaning System
In 2012, neuroscientist Maiken Nedergaard and her team at the University of Rochester made a pivotal discovery. They identified a previously unknown network in the brain that flushes out metabolic waste using cerebrospinal fluid (CSF) — the clear liquid that surrounds and cushions the brain and spinal cord.
Unlike the rest of the body, which relies on the lymphatic system to clear waste, the brain lacks traditional lymphatic vessels. Instead, the glymphatic system (a blend of “glial” cells and “lymphatic”) uses perivascular spaces — tunnel-like channels around blood vessels — to transport fluid. CSF enters these spaces, mixes with interstitial fluid (the fluid surrounding brain cells), picks up waste products like proteins, and then exits along veins to be removed from the brain.
The big surprise? This cleaning process is largely inactive during wakefulness but ramps up dramatically during sleep.
Why Sleep Supercharges Brain Cleansing
During wakefulness, brain cells are busy with thinking, learning, and processing information. This high activity keeps the spaces between cells tight, limiting fluid flow. But when you fall asleep — particularly during deep non-REM sleep — several changes occur:
- Brain cells shrink slightly, expanding the interstitial spaces by up to 60%.
- Blood flow decreases in certain areas, allowing more room for CSF to surge in.
- Slow, synchronized brain waves and coordinated neuronal activity generate rhythmic pulses that propel the fluid through the tissue.
This creates a wave-like “washing” effect: fresh CSF flows in, mixes with waste-laden fluid, and carries toxins out. Key waste products cleared include beta-amyloid and tau proteins, which accumulate in Alzheimer’s disease and impair neuron communication.
Studies in mice showed that glymphatic clearance is up to twice as efficient during sleep compared to wakefulness. Human imaging and recent research confirm similar patterns, with deep sleep providing the optimal conditions for this rinse cycle.
The Power Source: Neurons as Miniature Pumps
Recent studies have pinpointed exactly what drives this fluid movement. In 2024, researchers at Washington University School of Medicine discovered that neurons themselves act as “miniature pumps.” During sleep, they fire electrical signals in coordinated, rhythmic waves that physically push fluid through the dense brain tissue.
Additional 2025 research highlighted how norepinephrine (a neurotransmitter) pulses during non-REM sleep help regulate blood vessel dilation and fluid flow. Synchronized neural activity, brain waves, blood flow, and CSF movement all align like a well-orchestrated symphony to maximize waste removal.
Interestingly, if sleep is disrupted or insufficient, the brain may attempt compensatory “micro-cleaning” cycles even while awake — leading to brief lapses in attention and focus, as shown in 2025-2026 studies from MIT and others.
Implications for Brain Health and Disease
An efficient glymphatic system appears crucial for long-term brain health. Impaired clearance has been linked to buildup of toxic proteins, increasing risks for Alzheimer’s, Parkinson’s, and other dementias. Chronic sleep deprivation or poor sleep quality could accelerate this by reducing nightly cleaning.
On the positive side, prioritizing good sleep — aiming for 7-9 hours of quality rest, with plenty of deep sleep stages — supports this natural detox. Lifestyle factors like regular exercise, a healthy diet, and avoiding sleep disruptors (e.g., certain sleep aids that may interfere with fluid dynamics) can help optimize the process.
Sleep plays a pivotal role in memory consolidation — the process that stabilizes and strengthens newly formed memories, transforming fragile, short-term traces into more durable, long-term representations. While learning happens during wakefulness, consolidation largely occurs offline during sleep, when the brain replays experiences, integrates information, and prunes irrelevant details. Recent research (2024–2026) underscores that sleep isn’t just supportive — it’s essential, with mechanisms like neural replay, coordinated brain oscillations, and targeted reactivation driving the process.
How Memory Consolidation Works During Sleep
Memory consolidation involves two main types:
- Declarative memory (facts, events, episodic knowledge) — often hippocampus-dependent initially.
- Procedural memory (skills, habits, motor sequences) — more reliant on other brain areas like the basal ganglia and cortex.
During wakefulness, new information is encoded primarily in the hippocampus (for declarative memories). But the hippocampus has limited capacity for long-term storage. Sleep enables systems consolidation: memories are gradually redistributed to neocortical networks for permanent storage, becoming less dependent on the hippocampus.
A core mechanism is neuronal replay — the brain reactivates patterns of neural activity from recent experiences. In the hippocampus, this replay occurs during sharp-wave ripples (SWRs, brief high-frequency bursts ~80–200 Hz). These ripples coordinate with neocortical slow oscillations (0.5–1 Hz) and thalamocortical spindles (9–15 Hz) during non-REM sleep, creating “spindle-ripple events” that facilitate information transfer from hippocampus to neocortex.
This dialogue allows memories to integrate with existing knowledge, extract schemas or generalizations, and strengthen relevant connections via synaptic plasticity.
The Roles of Different Sleep Stages
Sleep cycles through non-REM (NREM) stages (light N1/N2, deep slow-wave sleep/SWS or N3) and REM sleep, each contributing uniquely:
- Slow-wave sleep (SWS/deep NREM) — Dominant for declarative and episodic memory consolidation. Hippocampal replay is prominent here, driven by slow oscillations that group reactivations. Studies show SWS-rich early-night sleep boosts fact/event recall and integration into semantic networks. Spindles and ripples nest precisely, promoting transfer and reducing interference.
- REM sleep — More linked to procedural, skill-based, and emotional memory. It supports integration of motor sequences, emotional processing (e.g., reducing emotional intensity of memories), and creative recombination. Late-night REM-rich periods enhance procedural improvements (e.g., mirror-tracing or texture discrimination).
Many theories converge: active systems consolidation (hippocampus-to-cortex transfer during SWS), dual-process (SWS for declarative, REM for procedural), and sequential (SWS tags/strengthens, REM integrates). Recent evidence favors complementary roles, with full cycles (SWS + REM) often yielding the strongest benefits.
Evidence from Recent Studies (2024–2026)
- Targeted Memory Reactivation (TMR) — Cues (sounds/odors) presented during sleep bias replay, enhancing specific memories (declarative, procedural, emotional). 2024 reviews highlight TMR’s noninvasive power to manipulate consolidation.
- Sleep restriction/deprivation — Impairs both encoding and consolidation. Short sleep matches total deprivation’s harm for some tasks; meta-analyses show declarative memory suffers most from SWS loss.
- Neural mechanisms — 2025–2026 work emphasizes oscillations, neuromodulators (e.g., low norepinephrine in NREM), and synaptic remodeling. Replay during SWS favors schema formation and future-relevant memories.
- Special cases — Children show more efficient declarative consolidation (abundant SWS); disruptions (e.g., sleep-disordered breathing) impair procedural overnight gains.
Practical Implications
Prioritizing 7–9 hours of quality sleep — especially with deep SWS and REM — boosts retention. Learning new material before bed leverages post-encoding consolidation. Naps (with SWS) aid declarative memory; full nights enhance complex integration.
Conversely, chronic poor sleep accelerates forgetting and raises risks for memory-related disorders (e.g., impaired clearance in Alzheimer’s links to reduced deep sleep).
In essence, sleep transforms daily experiences into lasting knowledge. It’s not passive rest — it’s active brain work, replaying yesterday to prepare for tomorrow.
The Takeaway: Sleep as Essential Maintenance
Far from being passive downtime, sleep is when your brain performs vital housekeeping. The glymphatic system’s nightly cleanse resets your brain chemistry, clears metabolic debris, and may protect against cognitive decline.
Next time you hit the snooze button, remember: you’re not just resting — you’re giving your brain its essential deep clean. Prioritizing sleep isn’t laziness; it’s one of the most powerful things you can do for lifelong brain health.
Sources include research from the University of Rochester, Washington University School of Medicine, Scientific American, and recent studies in journals like Nature and Cell (2024-2025).