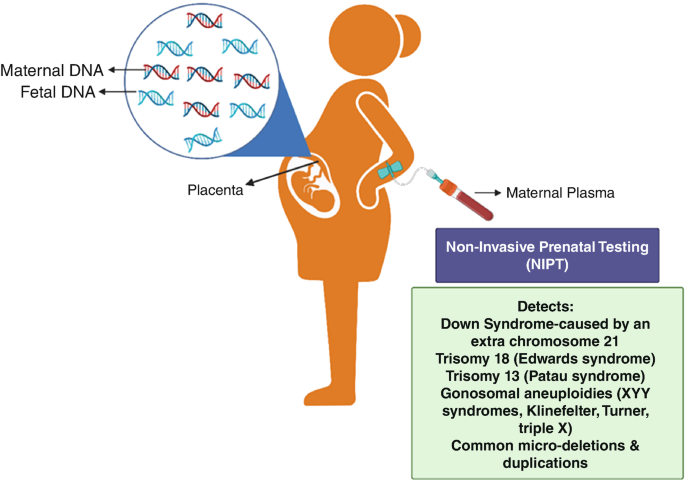
The journey of prenatal testing has been one of the most remarkable stories in modern medicine. For decades, the only way to obtain definitive genetic information about a growing fetus was through invasive procedures like amniocentesis or chorionic villus sampling (CVS)—tests that, while accurate, carried a small but real risk of miscarriage . The discovery of cell-free fetal DNA (cffDNA) in maternal blood in 1997 changed everything, ushering in the era of Non-Invasive Prenatal Testing (NIPT) . Today, NIPT is a routine screening tool that excels at detecting common chromosomal disorders like Down syndrome (trisomy 21) with over 99% accuracy .
However, the current generation of tests represents just the beginning. The future of prenatal diagnostics is poised for a quantum leap. Scientists are developing advanced blood tests that go far beyond screening for a handful of conditions. These emerging technologies promise to provide a comprehensive, safe, and early window into the fetal genome, potentially detecting hundreds of genetic diseases from a single tube of the mother’s blood.
The Current Landscape: A Foundation for the Future
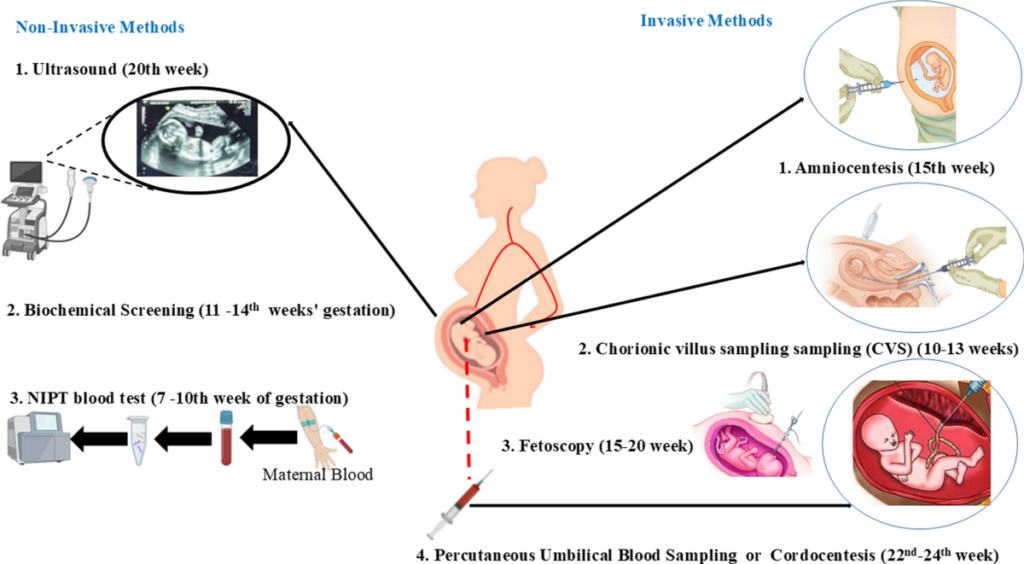
To appreciate the future, it is essential to understand the present. Current NIPT analyzes fragments of DNA shed from the placenta into the maternal bloodstream . It is a powerful screening tool, but it has limitations. Its primary focus is on whole-chromosome issues (aneuploidies) like trisomies 13, 18, and 21 . Its performance is less robust for other conditions, such as sex chromosome aneuploidies and microdeletion syndromes . Furthermore, because the fetal DNA is mixed with a larger amount of the mother’s own DNA, and because the placenta’s DNA can sometimes differ from the fetus’s (a phenomenon known as confined placental mosaicism), results are not always definitive and positive screens require invasive follow-up for confirmation .
The next wave of innovation aims to overcome these hurdles by harnessing new analytes, more powerful technologies, and deeper biological insights. Here are the most promising frontiers.
1. From DNA to RNA: Decoding the Body’s Instruction Manual
While DNA is the blueprint, RNA is the active instruction manual that carries out the blueprint’s commands. A new class of tests is moving beyond static DNA to analyze cell-free RNA (cfRNA) in maternal blood. This approach offers a dynamic view of fetal and placental health in real-time.
What it is: Unlike DNA, which is the same in every cell, RNA reflects which genes are actively being “expressed” or used. By analyzing cfRNA fragments circulating in the mother’s blood, scientists can gain insights into the function of the placenta and fetal organs .
What it can detect: The potential of cfRNA is vast. The company Mirvie has developed a platform that analyzes RNA patterns to predict complications like fetal growth restriction (FGR) as early as 17–22 weeks of gestation—well before it would be detected by traditional ultrasound . This platform has already shown promise in predicting preterm preeclampsia and spontaneous preterm birth .
In another groundbreaking application, researchers at the National University of Singapore have developed a cfRNA test that can assess fetal lung maturity. By measuring 21 specific RNA fragments, the test can determine if a fetus’s lungs are mature enough to breathe on their own, achieving 97-100% accuracy . This could revolutionize the management of threatened preterm birth by guiding doctors on whether to administer risky and potentially unnecessary steroids to accelerate lung development .
2. Comprehensive DNA Sequencing: Catching the Full Spectrum of Disease
The most significant leap forward in NIPT is the move toward comprehensive, genome-wide screening that can detect not just whole-chromosome issues, but also tiny mutations within individual genes.
The Technology: COATE-seq
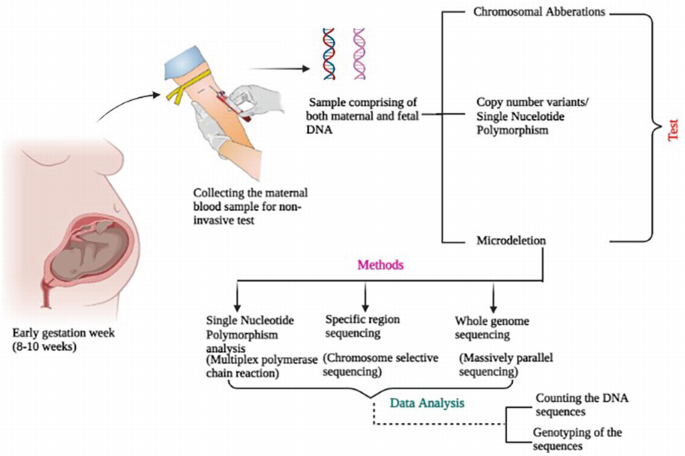
A revolutionary technique called “coordinative allele-aware target-enrichment sequencing” (COATE-seq) has been developed to address the fundamental challenge of separating the tiny amount of fetal DNA from the overwhelming background of maternal DNA . This method uses high-coverage, targeted sequencing to analyze read depth, allelic fractions, and linked single nucleotide polymorphisms simultaneously, allowing for an accurate reconstruction of the fetal genome.
What it can detect: The power of COATE-seq was demonstrated in a large prospective study of over 1,000 high-risk pregnancies. It successfully detected a broad range of genetic abnormalities in a single, non-invasive test, including:
- Common aneuploidies (trisomies 13, 18, 21) .
- Monogenic disorders: These are diseases caused by a mutation in a single gene, such as achondroplasia (a common cause of dwarfism), Noonan syndrome, and various skeletal dysplasias linked to genes like FGFR3, COL1A1, and PTPN11 .
The study found that COATE-seq identified pathogenic genetic variants in 135 pregnancies, a detection rate 60.7% higher than conventional cfDNA testing . This demonstrates that a single blood test could soon screen for hundreds of debilitating single-gene disorders that are currently not detected until after birth or through targeted invasive testing.
3. Metabolomics: Reading the Body’s Chemical Fingerprint
Moving even beyond genes and their expression, the future of prenatal testing may lie in metabolomics—the study of small molecules, or metabolites, that are the end products of cellular processes. These metabolites reflect the ultimate physiological state of the fetus and placenta.
What it is: Every genetic condition and physiological process creates a unique chemical footprint. By analyzing the metabolites in maternal blood or urine, researchers can identify patterns associated with specific fetal anomalies or genetic disorders .
What it can detect: A comprehensive literature review published in Prenatal Diagnosis highlights the potential of metabolomics as a powerful adjunct to genetic testing . For instance, it could help in the interpretation of “variants of uncertain significance” (VUS) found on genomic sequencing. If a genetic variant is detected, but its impact is unknown, an abnormal metabolic profile linked to that gene could provide the crucial evidence that the variant is indeed pathogenic. While still largely a research tool, metabolomics promises to add a functional layer to prenatal diagnosis, moving beyond what the genes say to what the body is actually doing.
4. Fetal Cells: The Ultimate, Elusive Prize
The “holy grail” of non-invasive prenatal diagnosis has always been the isolation and analysis of intact fetal cells circulating in the mother’s blood . While cffDNA consists of fragmented genetic material, a whole fetal cell contains a complete, undiluted copy of the fetal genome.
What it is: During pregnancy, rare fetal cells, such as nucleated red blood cells, cross the placenta and enter the maternal circulation . Capturing and analyzing these cells would provide a perfect sample for genetic testing, completely free from maternal contamination.
What it could detect: Access to intact fetal cells would allow for the definitive diagnosis of virtually any genetic condition—from single-gene disorders to complex chromosomal rearrangements—with the same accuracy as an invasive procedure, but without any risk. The challenge has always been the extreme rarity of these cells, making them incredibly difficult to find and isolate. However, drawing on lessons from oncology, where “liquid biopsies” are used to detect rare circulating tumor cells, researchers are developing ultra-sensitive technologies to finally make this a clinical reality .
The Road Ahead: Promise and Peril
The future of prenatal testing is one of unprecedented detail and scope. Within the next decade, a single blood test could potentially screen a pregnancy for:
- All major chromosomal abnormalities.
- Hundreds of severe single-gene disorders.
- Risk of later-in-pregnancy complications like preeclampsia and growth restriction.
- Functional assessments of fetal organ maturity.
However, this power comes with significant challenges. As highlighted by experts, the expansion of NIPT raises critical ethical, social, and policy questions . These include:
- Informed Consent: How do we counsel parents about the vast amount of information, including findings of “variants of uncertain significance” or adult-onset conditions, that such tests could reveal?
- Equitable Access: These advanced tests, particularly those using high-coverage sequencing, are expensive. Ensuring equitable access for all, regardless of socioeconomic status, will be a major challenge requiring policy interventions .
- Counseling Capacity: A healthcare system already struggling with genetic counselor shortages will need new frameworks to support clinicians and families in navigating complex genetic information .
- Regulatory Oversight: As technology races ahead, robust regulatory frameworks are essential to ensure clinical validity, reliability, and patient safety .
Conclusion
The journey from detecting a handful of chromosomal conditions to comprehensively screening the fetal genome from a simple blood test is well underway. With technologies like cfRNA analysis, COATE-seq, metabolomics, and fetal cell isolation on the horizon, the definition of “routine prenatal care” is set to be transformed. These innovations hold the immense promise of empowering parents and clinicians with knowledge earlier than ever before, enabling better preparation, targeted interventions, and improved outcomes for babies and families worldwide. The future of prenatal diagnosis is not just non-invasive; it is comprehensive, personalized, and profoundly insightful.

