Understanding Immunodeficiency: When a Child Cannot Produce IgG and IgM
Imagine a child who catches every cold that passes through the classroom, who struggles to recover from infections that other kids shake off in days, who seems to spend more time in doctors’ offices than on playgrounds. For some families, this isn’t imagination—it’s the reality of living with a child who has a primary immunodeficiency disorder that prevents the body from producing its own antibodies.
When a child cannot produce immunoglobulins IgG and IgM, the medical picture changes dramatically. These antibodies—specifically Immunoglobulin G (IgG) and Immunoglobulin M (IgM)—form the backbone of the immune system’s ability to fight infection. Without them, the child is essentially defenseless against bacteria and other invaders.
This article explains what happens when a child cannot produce these critical antibodies, why regular infusions of immunoglobulin are necessary, and what families need to know about this lifelong treatment journey.
The Immune System: A Quick Primer
To understand why IgG and IgM are so essential, we need to briefly look at how the immune system works.
The Five Immunoglobulins
Immunoglobulins (often called antibodies) are proteins produced by plasma cells (mature B-cells) that act as the body’s defense force. There are five main types:
| Immunoglobulin | Function | Location |
|---|---|---|
| IgG | Most abundant (75-80%); provides long-term immunity; crosses placenta to protect newborns | Blood, tissue fluids |
| IgM | First responder; produced during initial infection; activates complement system | Blood, lymphatic fluid |
| IgA | Protects mucosal surfaces (respiratory, digestive tracts) | Mucous membranes, saliva, tears |
| IgE | Involved in allergic responses and parasite defense | Tissues, bloodstream |
| IgD | Functions primarily on B-cell surfaces; role still being studied | B-cell surfaces |
The B-Cell Journey
B-cells (lymphocytes) are born in bone marrow and mature there. When they encounter an invader—a bacterium, virus, or fungus—they transform into plasma cells that produce antibodies specifically designed to fight that particular threat.
This process takes time. Initially, the body produces IgM antibodies as a rapid response. Later, it produces IgG antibodies for long-term protection and immunological memory—the reason vaccines work.
When a child cannot produce IgG and IgM, this entire defense system is compromised or completely absent.
The Underlying Conditions: Why Can’t a Child Produce These Antibodies?
Several primary immunodeficiency disorders can prevent a child from producing IgG and IgM. The most common include:
1. X-Linked Agammaglobulinemia (XLA)
XLA, also known as Bruton’s agammaglobulinemia, is the classic example of a condition where a child cannot produce antibodies.
What happens:
- A genetic mutation on the X chromosome (the BTK gene) prevents B-cells from maturing
- Without mature B-cells, the body cannot produce any immunoglobulins
- All five immunoglobulin types are severely reduced or absent
Who it affects:
- Almost exclusively males (since males have only one X chromosome)
- Symptoms typically appear around 4-8 months of age, when maternal antibodies (passed from mother during pregnancy) wear off
Key lab findings:
- IgG levels significantly below normal (often <200 mg/dL; normal for age: 400-1,200 mg/dL)
- IgM and IgA also severely decreased or undetectable
- Extremely low or absent B-cells in blood
2. Common Variable Immunodeficiency (CVID)
CVID is the most common symptomatic primary immunodeficiency requiring immunoglobulin therapy.
What happens:
- B-cells are present but fail to mature into antibody-producing plasma cells
- The exact genetic cause varies; multiple genes can be involved
- IgG levels are low, often accompanied by low IgA and/or IgM
Who it affects:
- Affects males and females equally
- Often diagnosed in childhood, but can present at any age
- May run in families
3. Autosomal Recessive Agammaglobulinemia (ARA)
Similar to XLA but caused by mutations in different genes (such as the μ heavy chain gene or Igα gene). Affects both males and females.
4. Hyper-IgM Syndrome
A group of disorders where:
- IgG and IgA are very low
- IgM is normal or elevated (hence the name)
- The immune system is stuck in “first response” mode and cannot switch to producing IgG
The Consequences: What Happens Without IgG and IgM?
When a child cannot produce IgG and IgM, the consequences are severe and predictable:
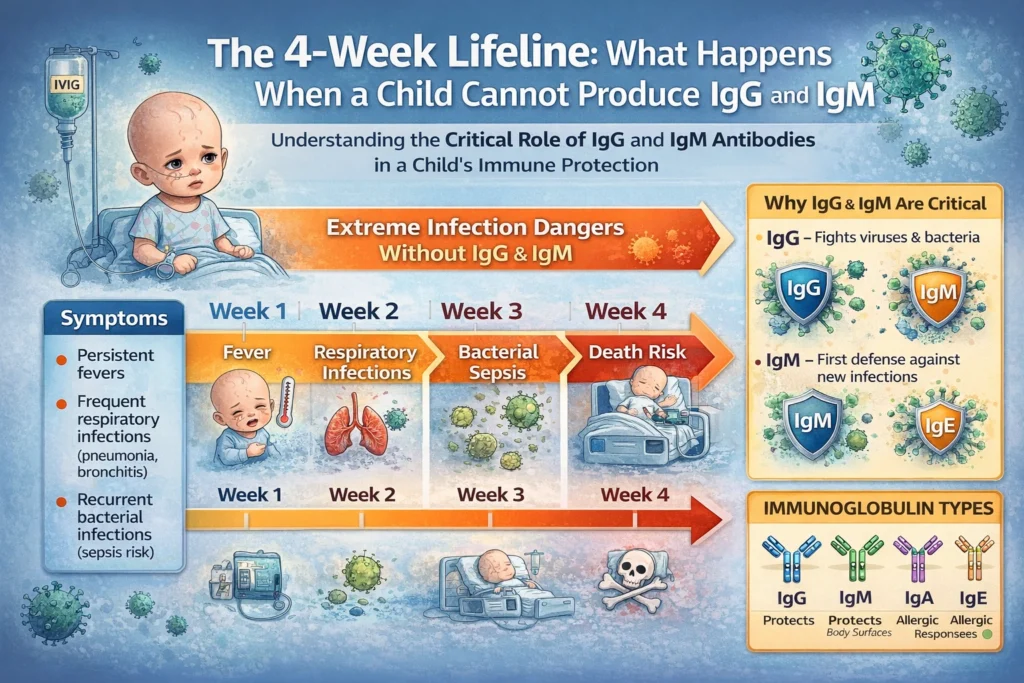
Recurrent, Severe Infections
Bacterial infections become relentless:
- Respiratory: Pneumonia, bronchitis, sinusitis, otitis media (ear infections)
- Gastrointestinal: Chronic diarrhea, giardiasis, other enteric infections
- Skin: Cellulitis, abscesses
- Systemic: Sepsis, meningitis
The hallmark is recurrent infections with encapsulated bacteria—organisms like Streptococcus pneumoniae, Haemophilus influenzae, and Staphylococcus aureus—which require antibodies for effective clearance.
Poor Response to Vaccines
When these children receive vaccines, their bodies cannot mount an antibody response. This is often a diagnostic clue—a child who has had all recommended vaccines but shows no protective antibody levels.
Failure to Thrive
The constant cycle of infection, inflammation, and recovery takes a toll. These children often struggle with:
- Poor weight gain
- Slowed growth
- Chronic fatigue
- Developmental delays
Chronic Complications
Without proper treatment, long-term damage accumulates:
- Bronchiectasis: Permanent dilation of airways from repeated lung infections
- Chronic sinus disease
- Hearing loss from recurrent ear infections
- Organ damage from disseminated infections
The Lifeline: Immunoglobulin Replacement Therapy
Since these children cannot produce their own antibodies, the treatment approach is elegantly simple in concept: give them antibodies from healthy donors.
What Is Immunoglobulin Replacement Therapy?
Immunoglobulin (often abbreviated as IG) is a medication made from the pooled plasma of thousands of healthy donors. It contains the full spectrum of antibodies—IgG (primarily), plus trace amounts of IgA and IgM—that a healthy immune system produces.
Think of it as a “passive immune system in a bag.” It doesn’t teach the child’s immune system to make its own antibodies, but it provides immediate protection against whatever infections the donors’ immune systems have encountered.
The 4-Week Schedule: Why This Timing?
The injection schedule—typically once every 4 weeks—is not arbitrary. It’s based on the half-life of IgG.
IgG half-life: Approximately 21-28 days
This means that after an infusion, the IgG level in the child’s blood gradually declines over about a month. By week 4, levels reach their lowest point (called the “trough level”). The next infusion brings them back up to protective levels.
The goal of therapy is to keep IgG trough levels above 500-800 mg/dL (depending on the child and clinical response). Below this, the risk of breakthrough infections increases significantly.
Two Administration Routes
1. Intravenous Immunoglobulin (IVIG)
What it is:
- Infused directly into a vein through an IV line
- Takes 2-4 hours per session
- Typically done in a hospital infusion center, home infusion setting, or specialized clinic
Frequency:
- Usually every 3-4 weeks
- The dose is calculated based on weight and trough levels
Advantages:
- One session covers the full dose
- No need for weekly administrations
- Medical supervision during infusion
Considerations:
- Requires venous access (can be challenging in small children)
- Travel to infusion site required
- Potential for systemic side effects (headache, fatigue, fever)
2. Subcutaneous Immunoglobulin (SCIG)
What it is:
- Infused under the skin (into subcutaneous tissue) using a small pump
- Takes 1-2 hours
- Can be done at home by trained family members
Frequency:
- Often weekly rather than monthly
- Smaller doses more frequently help maintain more stable levels
Advantages:
- Home-based therapy
- More stable IgG levels (fewer peaks and troughs)
- Fewer systemic side effects
Considerations:
- More frequent administration
- Requires family training and commitment
- Local site reactions (redness, swelling) are common
The Process: What Families Experience
For families with a child newly diagnosed with an immunodeficiency, the journey can feel overwhelming. Here’s what the treatment process typically involves:
Diagnosis Phase
- Suspicion: The child has recurrent, severe, or unusual infections
- Testing: Blood tests show low immunoglobulin levels (IgG, IgM) and poor vaccine responses
- Specialist referral: Pediatric immunologist takes over care
- Confirmatory testing: Flow cytometry (to check B-cell counts), genetic testing for specific mutations
Initial Treatment Phase
- Loading doses: Often more frequent infusions initially to achieve protective levels
- Establishing baseline: Regular monitoring to determine the child’s ideal trough level
- Finding the right product: Multiple immunoglobulin products exist; finding the one the child tolerates best may take time
Maintenance Phase
- Regular infusions: Every 3-4 weeks for IVIG, weekly for SCIG
- Ongoing monitoring: Trough levels checked every 3-6 months; dose adjusted as the child grows
- Infection surveillance: Prompt treatment of breakthrough infections
Common Concerns and Questions
Are There Side Effects?
IVIG side effects:
- During infusion: Headache, chills, fever, flushing, nausea
- Post-infusion: Fatigue, headache (sometimes severe), aseptic meningitis (rare)
- Long-term: Risk of thrombosis (rare); renal complications (very rare with modern products)
SCIG side effects:
- Local swelling, redness, itching at infusion sites
- Rarely, systemic side effects
Most side effects are manageable with pre-medications (acetaminophen, antihistamines, hydration) and rate adjustments.
Does the Child Still Have an Immune System?
Yes. Immunoglobulin replacement provides the antibodies the child cannot make, but the rest of the immune system—T-cells, phagocytes, complement system—still functions. The child may still have some ability to fight viruses and other pathogens, but bacterial protection depends entirely on the infusions.
Can They Live a Normal Life?
With consistent treatment and careful management, children with antibody deficiencies can:
- Attend school (with precautions during illness outbreaks)
- Participate in most activities
- Grow and develop normally
- Live into adulthood
However, vigilance is required. Infections that others fight off easily may require prompt medical attention.
The Outlook: Lifelong Treatment, Improving Outcomes
For children who cannot produce IgG and IgM, immunoglobulin replacement therapy is not a cure—it is a lifelong treatment. But it is a remarkably effective one.
Before the availability of immunoglobulin therapy in the 1950s and 1960s, children with XLA rarely survived past early childhood. Today, with regular immunoglobulin replacement, these children grow up, attend college, pursue careers, and have families of their own.
Advances on the Horizon
Research continues to improve outcomes:
- Gene therapy trials are exploring ways to correct the underlying genetic defects
- New immunoglobulin formulations with better tolerability and convenience
- Subcutaneous push techniques that reduce infusion time to 15-30 minutes
- Home-based monitoring allowing more personalized dosing
For Families: What to Ask Your Immunologist
If your child has been diagnosed with an antibody deficiency, these questions can help guide your care:
- “What is the specific diagnosis, and how does it affect my child’s immune system?”
- “What target trough level should we aim for, and how often will we check it?”
- “Is IVIG or SCIG better for our family’s situation?”
- “What signs of breakthrough infection should I watch for?”
- “What vaccinations are safe and recommended for my child?”
- “Are there any activities or exposures we should avoid?”
- “What is the plan for sick days, fevers, and urgent care?”
- “What support resources are available for our family?”
Summary: Understanding the 4-Week Cycle
| Component | Explanation |
|---|---|
| The problem | Child cannot produce IgG and IgM due to B-cell defect or maturation failure |
| The consequence | Susceptibility to recurrent, severe bacterial infections |
| The treatment | Immunoglobulin replacement from healthy donors |
| The timing | Every 3-4 weeks (based on IgG half-life of 21-28 days) |
| The goal | Maintain protective IgG trough levels (typically 500-800 mg/dL) |
| The outcome | Dramatically reduced infection risk; near-normal quality of life |
Final Thoughts
When a child cannot produce IgG and IgM, the immune system has a critical gap that cannot be filled by any medication except immunoglobulin replacement. The 4-week infusion schedule is not arbitrary—it is precisely calibrated to the biology of IgG, ensuring that protection never drops below the level needed to keep the child safe.
For families living this reality, the journey involves regular medical visits, careful infection monitoring, and the emotional weight of managing a chronic condition. But the reward is a child who can grow, learn, play, and thrive—protected by the generosity of thousands of blood donors who make this life-saving therapy possible.
If your child is showing signs of recurrent, severe, or unusual infections, speak with your pediatrician about the possibility of an immunodeficiency evaluation. Early diagnosis and treatment dramatically improve outcomes.
Medical Disclaimer: This article is for informational purposes only and does not constitute medical advice. Immunodeficiency disorders should be diagnosed and managed by qualified healthcare professionals, typically a board-certified immunologist. Treatment decisions should be made in consultation with your child’s medical team.