Coronary microvascular disease (CMD), also known as microvascular angina, small vessel disease, or cardiac syndrome X, is a serious but often invisible form of heart disease. It affects the tiny blood vessels (microvasculature) that branch off the larger coronary arteries and supply oxygen-rich blood deep into the heart muscle. Unlike classic coronary artery disease, where large arteries narrow due to plaque, CMD involves dysfunction, spasms, or impaired dilation in these microscopic vessels—reducing blood flow without any visible blockages on standard angiograms.

This condition earns the label “silent” because it frequently produces no obvious symptoms or only atypical ones that get dismissed as stress, menopause, or aging. It disproportionately strikes women, especially those over 40 entering perimenopause or menopause, when estrogen levels drop and protective effects on blood vessels fade. Many women live with it undiagnosed for years, facing heightened risks of heart attack, heart failure with preserved ejection fraction (HFpEF), stroke, and reduced quality of life.
Why It Strikes Women Over 40: The Estrogen Connection and Beyond
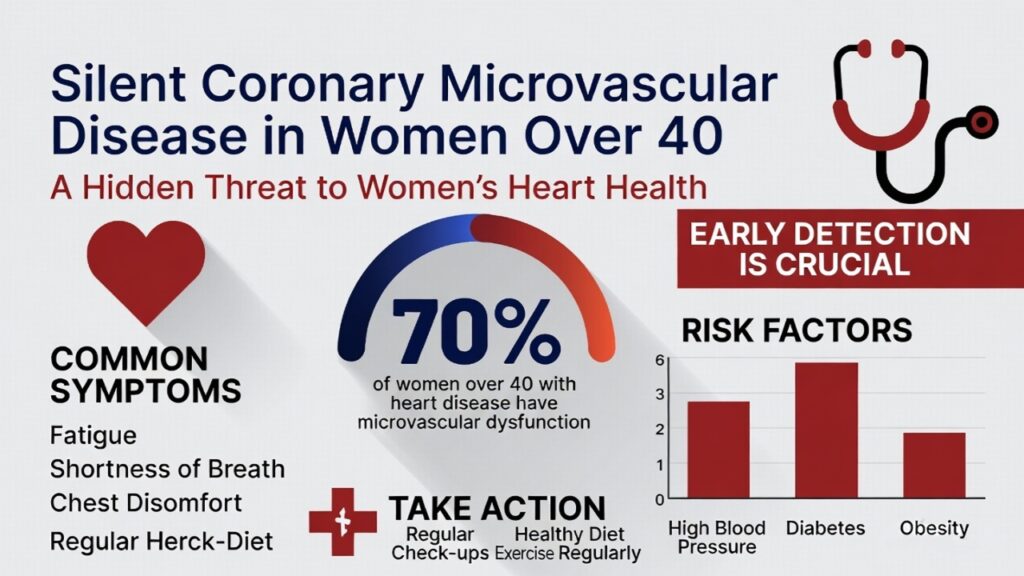
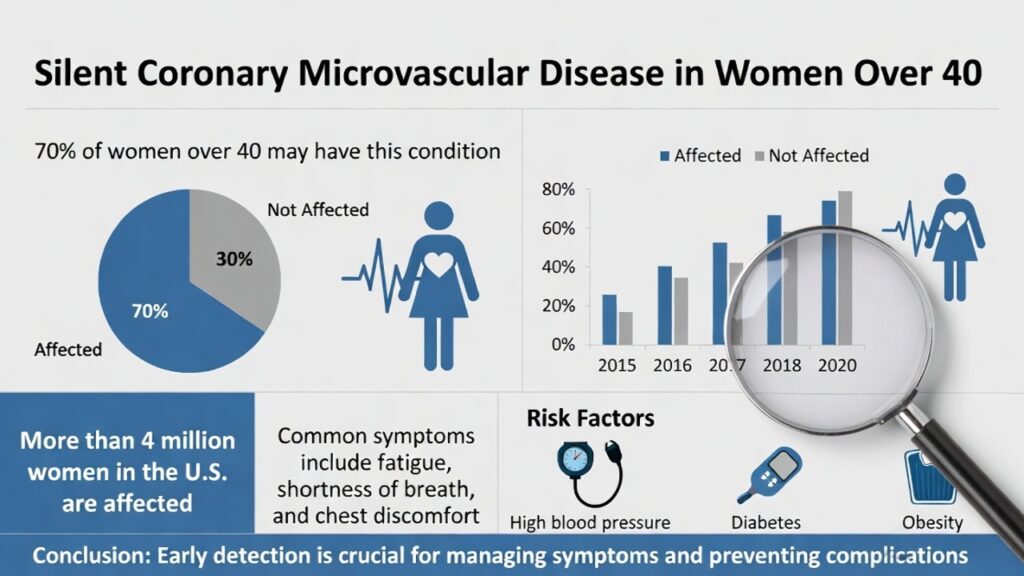
Women develop CMD far more often than men—accounting for up to 75% of cases in some studies. The shift typically accelerates after age 40 as estrogen declines during menopause. Estrogen helps keep small vessels flexible and responsive; its reduction leads to endothelial dysfunction (damage to the vessel lining), spasms, and poor blood-flow regulation.
Postmenopausal women also accumulate more traditional risk factors, including higher blood pressure, unfavorable cholesterol shifts, weight gain, and inflammation. Other contributors include:
- Diabetes or insulin resistance (women with diabetes face higher CMD risk than men)
- High blood pressure
- Obesity (BMI ≥30)
- Smoking or tobacco use
- Sedentary lifestyle
- Chronic stress or depression
- Family history (especially in women)
- Polycystic ovary syndrome or pregnancy complications (e.g., preeclampsia)
In one analysis of women with chest pain and no obstructive coronary arteries (a group called INOCA), roughly 41–50% had confirmed CMD. Prevalence rises with age and is especially common in the 40–60 age range.
The “Silent” Nature: Why It Goes Unnoticed
Many women with CMD experience silent ischemia—periods when the heart muscle lacks oxygen yet produces no chest pain or classic warning signs. A key study of women with CMD found that 39% experienced ambulatory silent ischemia episodes on monitoring, with 93% of those episodes occurring without symptoms. In contrast, healthy controls had zero episodes.
When symptoms do appear, they are often atypical and easily missed:
- Prolonged chest pressure, tightness, or burning that occurs at rest or during emotional stress (not just exertion)
- Shortness of breath
- Extreme fatigue or lack of energy
- Pain in the jaw, neck, back, shoulders, or upper abdomen
- Nausea, dizziness, or sleep disturbances
These can mimic anxiety, acid reflux, or menopausal symptoms. Women are also more prone to completely silent heart attacks, especially if they have diabetes, which dulls pain perception. Standard stress tests or angiograms frequently come back “normal,” leading to delayed diagnosis and repeated emergency visits.
Diagnosis: Why It’s Challenging and How It’s Done
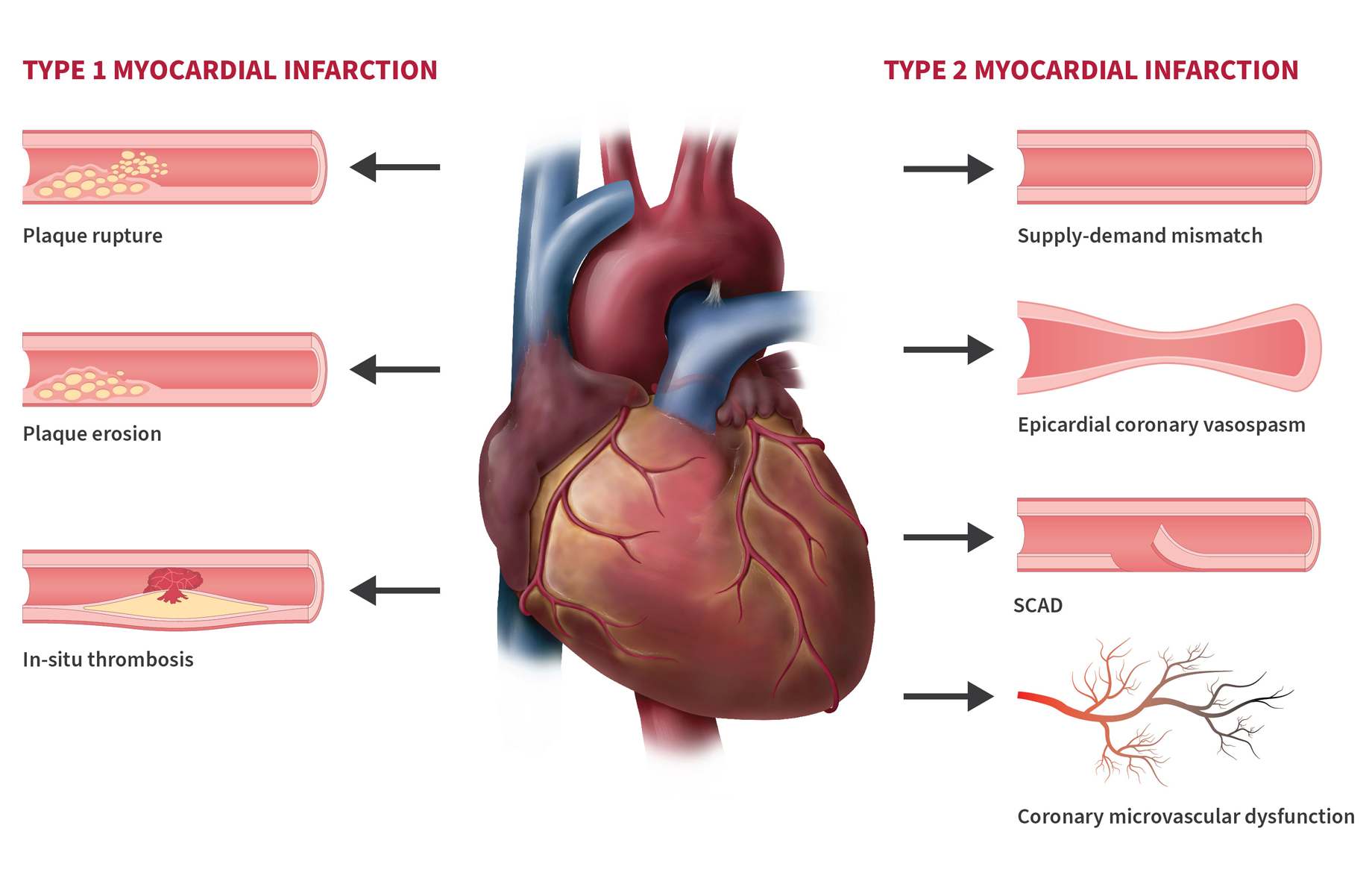
Traditional coronary angiography visualizes only the large arteries and misses microvascular issues. Diagnosis requires specialized testing when a woman over 40 has persistent symptoms despite “clear” arteries.
Modern approaches include:
- Invasive coronary function testing (gold standard): Measures coronary flow reserve (CFR) and index of microcirculatory resistance (IMR) during catheterization.
- Non-invasive imaging: Positron emission tomography (PET), cardiac MRI with perfusion, or advanced stress echocardiography.
- Ambulatory ECG monitoring to catch silent ischemia episodes.
Guidelines from the American Heart Association (AHA), American College of Cardiology (ACC), and European Society of Cardiology (ESC) now emphasize evaluating for CMD in women with INOCA symptoms. Early endotyping (identifying the exact mechanism—structural vs. functional) guides better management.
Treatment: Symptom Relief and Risk Reduction
There is no single cure, but targeted therapy dramatically improves quality of life and outcomes. Treatment is individualized:
- Lifestyle foundation: Heart-healthy Mediterranean-style diet, 150+ minutes of moderate exercise weekly, weight management, smoking cessation, stress reduction (mindfulness, yoga, support groups), and quality sleep.
- Medications:
- Beta-blockers or calcium channel blockers to reduce vessel spasms and heart workload
- Nitrates or ranolazine for persistent angina
- Statins and ACE inhibitors/ARBs to stabilize vessels and control risk factors
- Antiplatelet therapy (e.g., low-dose aspirin) in select cases
- Advanced options: Enhanced external counterpulsation (EECP) or, in research settings, targeted therapies addressing inflammation or endothelial dysfunction.
Clinical trials like CorMicA have shown that physiology-guided treatment (rather than standard care) significantly reduces angina and improves daily function within a year. Women who receive proper diagnosis and management see fewer hospitalizations and better long-term heart health.
Prevention: Empowering Women Over 40
The best defense is awareness and proactive steps:
- Know your risk—especially if you’re perimenopausal or have diabetes, hypertension, or a family history.
- Track symptoms: Don’t dismiss prolonged fatigue, unusual shortness of breath, or chest discomfort during rest or stress.
- Request advanced testing if standard evaluations are normal.
- Prioritize annual heart-health checks, including blood pressure, cholesterol, blood sugar, and BMI.
- Adopt protective habits early: regular physical activity, balanced diet, and stress management can preserve microvascular health.
Cardiac rehabilitation programs tailored for women are particularly effective for those already diagnosed.
Conclusion: From Invisible to Preventable
Silent coronary microvascular disease is not a “women’s problem” to ignore—it is a major, under-recognized driver of heart disease in women over 40. Thanks to improved awareness, specialized testing, and guideline updates, more women are now being diagnosed and effectively treated. The message is clear: chest pain (or even vague symptoms) with “normal” arteries is never normal. Talk to your doctor about microvascular disease if you’re over 40 and experiencing unexplained fatigue, breathlessness, or discomfort.
By recognizing the signs, advocating for the right tests, and committing to heart-healthy living, women can turn this silent threat into a manageable condition—and protect their hearts for decades to come. If you or someone you love fits this profile, schedule a heart check-up today. Early action saves lives.
Diagnostic Tests for Silent Coronary Microvascular Disease (CMD) in Women Over 40: An In-Depth Guide
Silent coronary microvascular disease (CMD), also called microvascular angina or INOCA (ischemia with no obstructive coronary arteries), is notoriously difficult to diagnose because the tiny vessels involved (smaller than 500 micrometers) cannot be seen on standard imaging. Classic tests like a regular ECG, exercise stress test, or standard coronary angiogram often return “normal” results despite ongoing ischemia. This leads to missed or delayed diagnoses, especially in women over 40, where symptoms may be vague (fatigue, shortness of breath, atypical chest pressure) or completely silent.
Modern diagnosis follows a stepwise approach guided by the 2021 AHA/ACC Chest Pain Guideline and ESC recommendations. It starts with ruling out obstructive disease and then moves to specialized tests that assess coronary flow reserve (CFR), microvascular resistance, and endothelial function. Below is a detailed breakdown of the key tests, how they work, what they measure, diagnostic cut-offs, strengths/limitations, and their role in clinical practice.
1. Why Standard Tests Miss CMD (and When to Escalate)
- Resting ECG and basic stress tests: May show no changes during silent ischemia episodes.
- Standard exercise treadmill test or nuclear stress test without advanced flow quantification: Often normal because they primarily detect large-artery blockages.
- Coronary angiography (invasive or CT): Visualizes only epicardial (large) arteries; microvessels are invisible. Up to 50–70% of women with persistent angina have “normal” angiograms yet proven CMD.
Escalation trigger: Persistent or atypical symptoms (especially at rest or with stress/emotion) in a woman >40 despite normal angiogram → proceed to advanced testing. Guidelines give a Class IIa recommendation for further evaluation in such patients.
2. Non-Invasive Tests (First-Line Screening Options)
These are preferred initially because they avoid catheterization. They detect impaired blood-flow reserve without visualizing the microvessels directly.
- Positron Emission Tomography (PET) Myocardial Perfusion Imaging with Myocardial Flow Reserve (MFR/MBFR)How it works: Uses radioactive tracers (e.g., ⁸²Rb or ¹³N-ammonia) injected at rest and during pharmacologic stress (adenosine, regadenoson, or dipyridamole). Dynamic imaging quantifies absolute myocardial blood flow (mL/min/g) in each heart region. Software calculates MFR = hyperemic flow / resting flow. Key metric & cut-off: MFR <2.0 is diagnostic for CMD (some labs use <2.5). Values <2.0 predict higher risk of heart events even without obstructive disease. Strengths: Most validated non-invasive method; provides global and regional quantification; reproducible; strong prognostic value (worse outcomes if resting flow is high, which is more common in women). Limitations: Radiation exposure, limited availability, expensive, requires caffeine abstinence 24–48 hours prior. Guideline role: Class IIa recommendation (AHA/ACC). Excellent first-line for suspected INOCA.
- Cardiac Magnetic Resonance (CMR) Imaging with Myocardial Perfusion Reserve (MPR/MPRI)How it works: First-pass perfusion imaging during rest and adenosine stress. Gadolinium contrast tracks blood flow through the myocardium. Semi-quantitative (upslope ratio) or fully quantitative methods generate MPR. Key metric & cut-off: Quantitative MPR <2.19 or semi-quantitative MPRI <1.84 indicates CMD. MPRI ≤1.47 is a strong predictor of major adverse events. Strengths: No radiation; excellent for tissue characterization; useful in MINOCA (heart attack with normal arteries); can detect silent ischemia. Limitations: Requires specialized sequences and post-processing; motion artifacts possible; not as widely available as PET. Guideline role: Class IIa (AHA/ACC); particularly helpful in women and for risk stratification.
- Stress Echocardiography with Coronary Flow Velocity Reserve (CFVR)How it works: Transthoracic Doppler ultrasound measures diastolic flow velocity in the mid-distal left anterior descending (LAD) artery at rest and during stress (exercise or adenosine). CFVR = stress velocity / rest velocity. Key metric & cut-off: CFVR <2.0–2.25 abnormal (lower values predict events in women with INOCA). Strengths: Widely available, no radiation, real-time imaging. Limitations: Operator-dependent; limited to LAD territory; technically challenging in some patients. Guideline role: Class IIb (less preferred than PET/CMR).
Additional non-invasive tool: 24–48-hour ambulatory (Holter) ECG monitoring to capture silent ST-segment changes during daily activities — useful when symptoms are absent but suspicion remains high.
3. Invasive Coronary Function Testing — The Gold Standard
Performed during cardiac catheterization (usually outpatient) when non-invasive tests are inconclusive or symptoms are severe/persistent. Takes 30–60 extra minutes after standard angiography. Safe (major complication rate <0.5–1%) when done by experienced operators.
Core measurements (performed in one or more major coronary arteries):
- Coronary Flow Reserve (CFR) Measures the microvasculature’s ability to increase blood flow 2–5 times above baseline. How measured:
- Thermodilution (most common, using PressureWire X): Saline boluses at rest and during hyperemia (adenosine IV 140 µg/kg/min); CFR = resting transit time / hyperemic transit time.
- Doppler wire (FloWire): Average peak velocity ratio (hyperemic/rest). Cut-off: <2.0 (Doppler) or <2.5 (thermodilution) = abnormal CMD. Formula (thermodilution):
- Index of Microcirculatory Resistance (IMR) Specific for microvascular health (unaffected by epicardial disease). How measured: During maximal hyperemia, IMR = distal coronary pressure (P_d) × hyperemic mean transit time. Cut-off: IMR ≥25 = elevated resistance (structural CMD). Formula:IMR=Pd×Tmn (hyperemia)(Normal <25; higher values indicate poor microvascular dilation.)
- Hyperemic Microvascular Resistance (HMR) (Doppler alternative): P_d / hyperemic average peak velocity; abnormal >2.4–2.5.
Acetylcholine (ACh) Provocation Testing (endothelium-dependent assessment)
- How performed: Incremental intracoronary ACh boluses or infusions after CFR/IMR. Monitors for symptoms, ECG changes, and vessel diameter/flow.
- Diagnoses:
- Epicardial spasm: ≥90% narrowing + chest pain + ischemic ECG.
- Microvascular spasm: Symptoms + ECG changes without epicardial narrowing.
- Endothelial dysfunction: <50% increase in blood flow or paradoxical constriction.
- Why crucial: Identifies functional vs. structural CMD and guides therapy (e.g., calcium-channel blockers for spasm).
Endotype classification (COVADIS criteria, used in guidelines): Combines results to pinpoint the exact mechanism (microvascular dysfunction, microvascular spasm, epicardial spasm, or mixed). This “precision diagnosis” improves outcomes, as shown in the CorMicA trial.
Procedure safety notes: Withhold caffeine/vasodilators 24–48 hours. Low risk when protocols are followed.
4. Current Guidelines & Diagnostic Pathway (2021–2025)
- AHA/ACC (2021 Chest Pain Guideline): Class IIa for invasive function testing or advanced non-invasive (PET/CMR) in persistent symptoms + non-obstructive CAD.
- ESC: Class I (Level B) for guidewire-based CFR/IMR in symptomatic patients with normal/mildly diseased arteries.
- Typical pathway for women >40:
- History + risk assessment.
- Rule out obstructive CAD (angiography or CT).
- Non-invasive CFR/MPR if available.
- Invasive comprehensive testing (CFR + IMR + ACh) if symptoms persist — now considered cost-effective and symptom-improving.
Special Notes for Silent CMD in Women Over 40
Women often have functional (endothelial) rather than structural CMD, higher resting flow, and more microvascular spasm. Silent ischemia is common (up to 39% on monitoring). Estrogen decline after 40 reduces vessel flexibility, making advanced testing especially valuable. Early diagnosis via these tests reduces emergency visits, improves quality of life, and lowers long-term risks of heart failure and events.
Bottom line: If you’re a woman over 40 with unexplained fatigue, breathlessness, or “normal” test results, ask your cardiologist about CFR, IMR, or PET/CMR evaluation. These targeted tests turn an invisible threat into a treatable condition. Always discuss with a specialist experienced in INOCA/CMD — early precision diagnosis saves hearts.