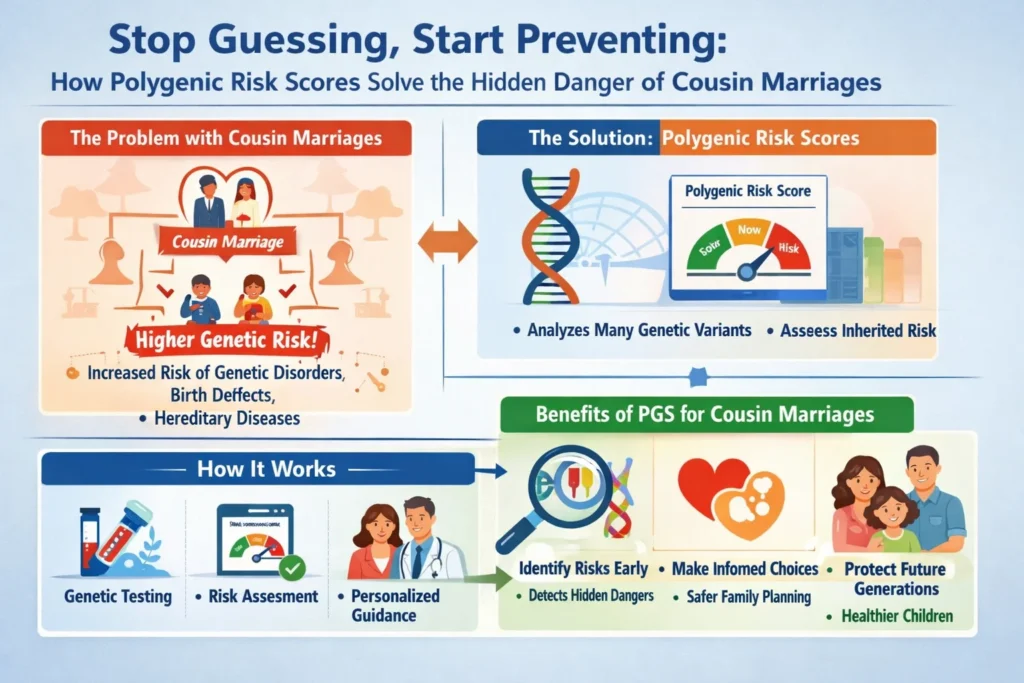
You’ve heard the warning a hundred times: marrying your cousin increases the risk of rare, devastating recessive disorders. So you did the responsible thing. You went for carrier screening. The panel came back clear. No cystic fibrosis. No Tay-Sachs. No spinal muscular atrophy. You exhaled. The danger, you assumed, was over. But here is what no genetic counselor told you: you just looked for a few needles in a haystack while ignoring the condition of the hay itself.
The hidden danger of consanguineous marriages isn’t just the rare mutation that shuts off a single gene. It’s the thousands of tiny, common genetic variants—harmless on their own but dangerous in duplicate—that accumulate when both parents share a recent ancestor.
These variants don’t cause a catastrophic disease in childhood. They quietly raise the lifetime risk of diabetes, heart disease, schizophrenia, and autoimmune disorders by 30%, 50%, or even 100%. And until now, you had no way to measure them. That ends today. Welcome to the era of polygenic risk scores—the precision tool that finally lets consanguineous families stop guessing about their children’s genetic future and start preventing disease before it ever begins.
Beyond Family Ties: Can Polygenic Risk Scores Redefine Counseling in Consanguineous Marriages?
For centuries, the family tree in many cultures has not so much branched out as woven itself into a tight, intricate lattice. In regions spanning North Africa, the Middle East, Central Asia, and parts of Southern Europe, consanguineous marriages—unions between second cousins or closer—remain a deeply embedded social cornerstone. These unions are rarely about genetic fatalism; they are about preserving wealth, solidifying tribal alliances, simplifying matchmaking, and ensuring a shared value system. Yet, when a genetic counselor looks at a pedigree chart with double lines connecting first cousins, a familiar chill runs down the spine.
The conventional narrative is stark and binary: consanguinity increases the risk of autosomal recessive disorders. The logic is impeccable. When parents share a recent common ancestor, their offspring have a higher probability of inheriting two identical copies of a harmful variant, known as a recessive pathogenic mutation. The result can be devastating—cystic fibrosis, spinal muscular atrophy, metabolic storage diseases, and profound intellectual disability.
But what if this binary narrative is incomplete? What if the greatest genetic risk of cousin marriage isn’t a single, rare, catastrophic mutation, but the accumulation of thousands of tiny, common genetic variations that, together, shape our destiny? This is where the revolutionary concept of the polygenic risk score (PRS) enters the conversation. For the first time, we have a tool that looks beyond the Mendelian boogeyman and asks a more nuanced, uncomfortable, and potentially transformative question: Beyond family ties, can polygenic risk scores redefine counseling in consanguineous marriages by predicting the risk of common, complex diseases like diabetes, schizophrenia, or heart disease?
To answer that, we must first dismantle a few old paradigms and then rebuild a new, culturally competent framework for the 21st century.
The Blind Spot of Classic Genetic Counseling
Traditional preconception and prenatal counseling for consanguineous couples follows a well-trodden path. The counselor draws a pedigree, calculates the inbreeding coefficient (F), and typically recommends carrier screening for a panel of recessive disorders prevalent in the couple’s ethnic group. The conversation is dominated by numbers: a first-cousin union carries an approximately 2-3% increased absolute risk of a serious congenital anomaly or recessive disease above the general population baseline of 2-3% (totaling 4-6%). The advice is often a binary choice: proceed, but with prenatal diagnosis; consider gamete donation; or avoid the union altogether.
This approach, while scientifically sound, suffers from three critical blind spots.
First, it is largely retrospective. It focuses on known, severe, early-onset diseases that are rare in the general population. It tells a couple nothing about their child’s lifelong risk for common, complex conditions like type 2 diabetes, hypertension, bipolar disorder, or coronary artery disease—the very diseases that fill hospital wards and shorten lifespans in middle age.
Second, it is culturally deaf. By framing the conversation almost exclusively around risk and pathology, counselors often alienate the very people they aim to help. Consanguinity is not a pathology; it is a kinship structure. In many societies, refusing a cousin’s hand in marriage can be a family betrayal. When science offers only a message of “higher risk” without actionable nuance, it is easily dismissed as Western bias.
Third, it ignores the vast, uncharacterized landscape of the genome that lies outside the exome. The old model assumes that “risk” equals “recessive mutation.” But what about additive genetic effects? What about the thousands of single nucleotide polymorphisms (SNPs), each with a tiny effect size, that cumulatively push a person toward or away from a disease state?
Enter the polygenic risk score.
What is a Polygenic Risk Score, Really?
Imagine a dimmer switch, not a light switch. A Mendelian mutation is a light switch: flip it (inherit two bad copies), and the light of disease turns on. A polygenic risk score, however, is a dimmer switch composed of thousands of tiny dials. Each dial represents a common genetic variant. Individually, none of them can cause disease. But when you sum up the effect of all the dials—some turned slightly up, others slightly down—you get a score. That score predicts the probability that a person will develop a given complex trait, from height to heart failure.
A PRS is calculated by aggregating the effects of millions of SNPs identified in massive genome-wide association studies (GWAS). For example, a high PRS for breast cancer doesn’t mean you have a BRCA mutation; it means your collection of common variants places you in the 90th percentile of genetic risk. Your lifetime risk might be 15% instead of the population average of 12%—a subtle but statistically significant shift.
Here is where the intersection with consanguinity becomes electrifying. While consanguinity increases the risk of identical-by-descent (IBD) segments for rare recessive mutations, it also increases genome-wide homozygosity. That is, offspring of cousin marriages have longer stretches of the genome where both chromosomes are identical, inherited from a common ancestor. This means that not only are they more likely to get two bad copies of a rare recessive gene, but they are also more likely to be homozygous (having two identical copies) at thousands of common SNP locations.
Why does homozygosity matter for PRS? Because for complex diseases, having two identical copies of a risk allele (homozygous) often confers a different effect than having one copy (heterozygous).
More importantly, the total genetic burden of a disease is not just the sum of risk alleles, but the additive effect of homozygosity across the genome. Studies in isolated, consanguineous populations have shown that increased runs of homozygosity (ROH) are associated with lower cognitive performance, shorter stature, and higher risk of schizophrenia—even after controlling for known recessive mutations.
This is the signature of polygenic homozygosity burden.
The Hypothesis: A New Layer of Risk
Let us formulate a testable, provocative hypothesis: In consanguineous marriages, the increased risk of common complex diseases is not solely, or even primarily, driven by rare recessive mutations. Instead, it is driven by the polygenic load of homozygosity—the amplification of thousands of small-effect risk variants that become more potent when present in two identical copies.
Consider type 2 diabetes. Over 500 common SNPs have been associated with its risk. A person’s PRS for diabetes is the sum of these risk alleles. In an outbred population, the distribution of this PRS follows a normal curve. But in an offspring of a first-cousin marriage, the genome contains longer stretches of homozygosity.
If, within those stretches, there are multiple risk-increasing alleles for diabetes, the child will be homozygous for them. And homozygosity can have a multiplicative, not just additive, effect on gene expression. A risk variant that in a single copy raises diabetes risk by 5% might, in two copies, raise it by 12% due to dosage effects or altered regulatory networks.
Therefore, a child from a consanguineous union might have a PRS that is not just the sum of inherited risk alleles, but a weighted sum where homozygosity at key loci creates a higher effective score. This means that current PRS models, which are trained primarily on outbred European populations, systematically underestimate the true polygenic risk in consanguineous families. We are not just missing the recessive bombs; we are missing the slow-burning polygenic fires.
The Cultural and Psychological Chasm
Now, imagine you are a genetic counselor sitting across from a young couple—let us call them Ahmed and Fatima—who are first cousins from a Moroccan Berber community. They have no family history of cystic fibrosis or Tay-Sachs. Their carrier screening panel is clear. They have come for preconception counseling, expecting a green light. Instead, you want to discuss polygenic risk scores for schizophrenia, coronary artery disease, and type 2 diabetes.
How do you begin?
The first challenge is epistemic trust. In many collectivist cultures, the concept of statistical risk is foreign. Decisions are made based on precedent, family history, and divine will, not on a percentile score from a GWAS. Introducing a PRS as a “score” implies a ranking, a judgment. “You have a high PRS for depression” sounds like a character flaw, not a probabilistic forecast.
Second, there is the problem of actionability. For a lethal recessive disease like Tay-Sachs, a positive finding leads to clear actions: PGD, prenatal diagnosis, or termination. For a high PRS for hypertension, what do you do? Counsel lifestyle modification? That is the same advice you would give to anyone. The lack of a decisive medical intervention makes the PRS feel like a surveillance tool without a purpose.
Third, consider the psychological burden of genomic fatalism. In an outbred context, a high PRS is a nudge. In a consanguineous context, if a couple is told, “Because you are cousins, your child’s PRS for schizophrenia is likely to be in the 80th percentile,” they may interpret this as an inescapable curse. It could stigmatize consanguinity even further, driving the practice underground rather than bringing it into the light of informed consent.
And yet, there is a counterargument. What if knowledge is power? What if a high PRS for diabetes motivates a family to adopt a traditional, low-glycemic diet? What if a high PRS for thrombosis prompts earlier anticoagulant prophylaxis during pregnancy? The promise of PRS is not genetic determinism; it is precision prevention.
Redefining Counseling: From Risk Aversion to Resilience Building
To integrate polygenic risk scores into counseling for consanguineous marriages, we must abandon the paternalistic, pathology-focused model of the 20th century. We need a new paradigm—let us call it Genomic Kinship Counseling (GKC) —built on four pillars.
Population-Specific PRS Calibration
The single greatest technical barrier is that PRS are notoriously portable. A score derived from a UK Biobank cohort of European ancestry performs poorly when applied to a Pakistani, Saudi, or Turkish population. Why? Because linkage disequilibrium patterns, allele frequencies, and environmental interactions differ. A variant that is protective in Europeans might be neutral or even harmful in a consanguineous Middle Eastern population due to different background homozygosity.
Therefore, the first step is massive, coordinated efforts to build consanguinity-aware PRS databases. This requires GWAS of complex diseases specifically in populations where first-cousin unions exceed 20-30% (e.g., Saudi Arabia, Qatar, parts of India). Only when we have PRS trained on homozygous blocks can we begin to generate accurate scores for these families. Without this, we are flying blind.
Dynamic, Narrative-Based Counseling
Forget the whiteboard and the Punnett square. Counseling for PRS must be narrative and visual. Use dynamic risk calculators that show a range of outcomes, not a single number. Explain that a PRS is like a weather forecast: “There is a 40% chance of rain” does not mean it will rain, but you might carry an umbrella. The umbrella, in this case, is lifestyle, monitoring, and early intervention.
Crucially, reframe the conversation from risk to resilience. Show Ahmed and Fatima that even with a high PRS for a disease, protective factors—diet, exercise, community support, even other genetic variants—can lower the actual outcome. The goal is not to scare them away from marriage but to equip them with a lifelong roadmap of proactive health surveillance for their future children.
The Ethical Integration of Psychosocial Support
Polygenic risk scores can cause anticipatory anxiety and familial guilt. A mother might blame herself for passing on a “bad” set of SNPs. A father might feel his lineage is tainted. Counseling must be paired with culturally adapted psychosocial support. This includes:
- Genograms that map not just disease but resilience factors (e.g., longevity, mental health coping strategies).
- Decision aids that separate the concept of genetic risk from moral worth.
- Community education that normalizes the fact that everyone has high PRS for something. Cousin marriage does not create risk; it redistributes and amplifies background risk.
Returning to the Individual—The Possibility of Somatic PRS
One of the most revolutionary, under-discussed angles is this: polygenic risk scores are not just for embryos. They can be calculated for living individuals. In a consanguineous marriage where a couple already has one child, you could calculate that child’s PRS for 10-20 common diseases. The result would not be a reason to love that child less, but a personalized health plan.
“Your daughter’s PRS suggests a higher likelihood of developing autoimmune thyroiditis by age 40. Let’s monitor her thyroid antibodies every two years starting at age 20.” This is not eugenics; it is preventive medicine. For consanguineous families who worry endlessly about unknown recessive risks, a PRS provides known, manageable risks. It replaces dread with data.
The Objections: Scientific, Ethical, and Cultural
No discussion of PRS in consanguineous settings is complete without addressing the formidable objections.
The Scientific Objection: PRS have low predictive power. For most diseases, the area under the curve (AUC) is 0.6-0.7, only marginally better than flipping a coin. Why introduce a weak predictor into high-stakes counseling? Response: Because weak predictors, aggregated across thousands of variants and adjusted for homozygosity, become strong population-level predictors. And for a couple making a once-in-a-lifetime decision, even a small, actionable signal is valuable.
The Ethical Objection: This will lead to a slippery slope toward prenatal selection against polygenic traits, including non-disease traits like educational attainment or height. In consanguineous communities, which may already face discrimination, this could exacerbate genetic stratification. Response: A valid point. Therefore, PRS counseling must be strictly limited to high-penetrance, medically actionable diseases (e.g., breast cancer, type 1 diabetes, familial hypercholesterolemia). A moratorium on PRS for behavioral or cosmetic traits is essential.
The Cultural Objection: You are imposing a Western, individualistic, biomedical framework on communities that view health holistically—as a balance of spiritual, social, and physical factors. Response: This is the most powerful objection.
The solution is not to abandon PRS but to co-design counseling protocols with community elders, religious scholars, and traditional healers. In Islamic jurisprudence, for example, preventive medicine is encouraged (tadawi). A PRS could be framed not as a threat but as a form of sadaqah (charitable foresight) to protect future generations.
A Case Study in Imagination
Let us imagine a future—say, 2035. A clinic in Dubai offers “Genomic Family Planning.” A couple, second cousins from a Bedouin lineage, walks in. They do not want carrier screening alone. They want the full picture. The counselor draws their blood, sequences their genomes, and calculates two things: (1) a traditional recessive mutation panel, and (2) a consanguinity-adjusted polygenic risk score for 12 common diseases.
The recessive panel is clear. But the PRS shows that their offspring have a predicted 1.6x higher risk of developing type 2 diabetes by age 50 compared to the general population, driven by homozygosity at four key loci. The counselor does not say, “Don’t have children.” She says, “Your child will have a genetic predisposition to insulin resistance. If you start intermittent fasting and a low-glycemic diet from childhood, you can reduce that risk by 40%. Also, there is a new continuous glucose monitor that can detect prediabetes a decade early.”
The couple leaves not with fear, but with a plan. They proceed with the marriage, have healthy children, and enroll those children in a preventive lifestyle program. Twenty years later, their children have normal metabolic health. The polygenic risk did not become destiny because it was illuminated in time.
Conclusion: Beyond Family Ties
To return to our opening question: Beyond family ties, can polygenic risk scores redefine counseling in consanguineous marriages?
The answer is a qualified, cautious, hopeful yes. But only if we are willing to do the hard work. We must build population-specific PRS models that account for homozygosity. We must train a new generation of counselors who are fluent in both Bayesian statistics and cultural humility. We must engage in transparent ethical deliberation about what risks are appropriate to disclose. And we must never forget that behind every pedigree chart is a family seeking love, continuity, and meaning—not just a probability.
The old narrative of consanguinity was a story of hidden bombs—rare, recessive, catastrophic. The new narrative, powered by polygenic risk scores, is a story of landscapes—gentle hills and valleys of risk, navigable with good maps and steady feet.
It replaces fatalism with foresight, stigma with strategy. In doing so, it honors the profound truth that family ties are not just chains of shared DNA; they are also the strongest protective factors we have. And now, for the first time, we have the genomic tools to ensure that those ties lead to health, not harm.
The double line on the pedigree need not be a warning sign. With the wise application of polygenic risk scoring, it can become a bridge between tradition and precision medicine—a line that connects not just cousins, but past, present, and future in a continuous, informed, and resilient embrace.