In the evolving landscape of preventive healthcare, oxidative stress biomarkers are emerging as a powerful frontier in anti-aging and disease prevention. As Americans increasingly prioritize healthspan—the number of years lived in good health—over mere lifespan, understanding and tracking oxidative stress offers a proactive way to detect “silent” aging processes early and intervene with lifestyle, diet, or targeted therapies.
What Is Oxidative Stress?
Imagine what happens to an apple slice when left exposed to air—it turns brown. Or think about iron rusting over time. A similar process occurs inside our bodies, and scientists call this oxidative stress .
At its core, oxidative stress is simply an imbalance between two opposing forces :
- Free radicals (reactive oxygen species, or ROS): Unstable molecules that damage cells, proteins, and DNA by stealing electrons
- Antioxidants: The body’s defense system that neutralizes free radicals
Think of free radicals as sparks flying from a campfire. A few sparks are normal and harmless. But when too many sparks land on surrounding grass, you need firefighters to prevent a blaze. Antioxidants are those firefighters .
The Oxygen Paradox
Here’s an interesting twist: oxygen, essential for life, also creates these damaging free radicals with every breath we take. In 1995, researcher Kelvin Davies coined the term “The Oxygen Paradox” to describe this phenomenon .
“Oxidative stress is what causes iron to rust and apples to go brown. For humans, it’s also part of the process that causes wrinkles, many diseases and even aging” .

Why Does Oxidative Stress Matter for Aging?
Oxidative stress accelerates aging through several mechanisms. It damages three critical components of our cells :
- DNA: Causes mutations and genetic instability
- Proteins: Impairs their normal function
- Lipids (fats): Damages cell membranes through a process called lipid peroxidation
This cellular damage accumulates over time and drives the hallmarks of aging, including mitochondrial dysfunction, chronic inflammation, and cellular senescence (when cells stop dividing but don’t die—they just linger and cause trouble) .
Diseases Linked to Oxidative Stress
When oxidative stress overwhelms the body’s defenses, it doesn’t just cause general aging—it contributes to specific chronic diseases :
| Disease Category | Specific Conditions |
|---|---|
| Cardiovascular | Atherosclerosis, high blood pressure, heart disease |
| Neurodegenerative | Alzheimer’s disease, Parkinson’s disease |
| Metabolic | Diabetes, insulin resistance |
| Inflammatory | Rheumatoid arthritis, inflammatory bowel disease |
| Other | Cancer, premature aging syndromes |
The Good News: A Little Stress Can Be Beneficial
Surprisingly, not all oxidative stress is bad. Low-level oxidative stress actually strengthens our cells through a process called hormesis—what doesn’t kill you makes you stronger .
When exposed to mild stress, cells:
- Slow their growth to conserve energy
- Activate protective genes
- Produce more antioxidant enzymes
- Repair damage more efficiently
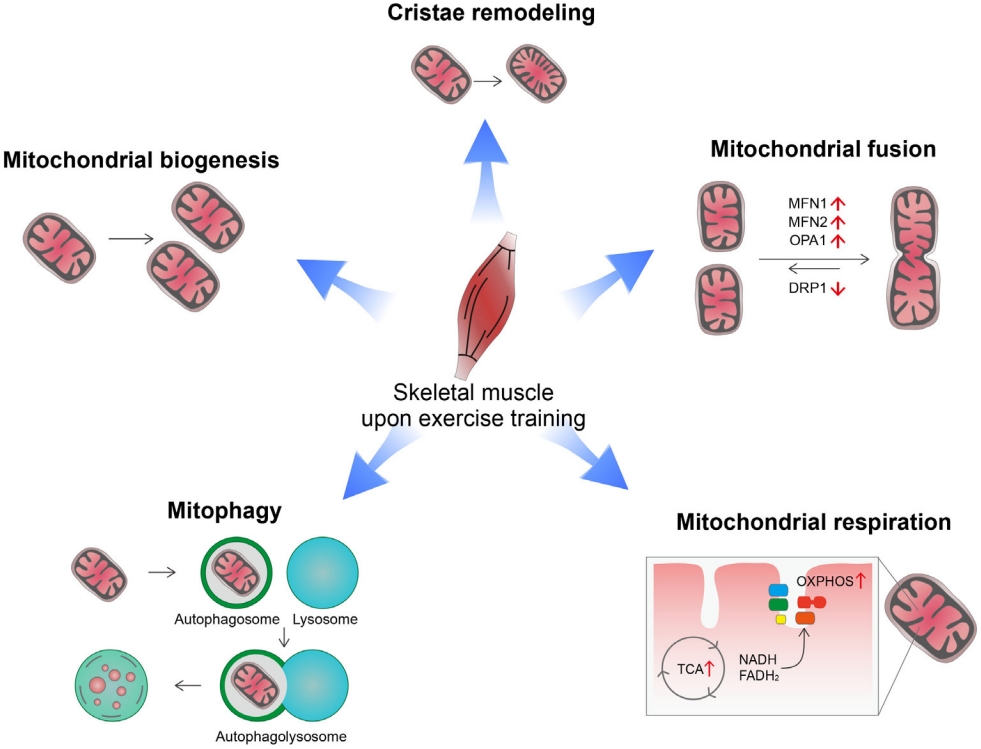
This is why moderate exercise is beneficial—it creates temporary oxidative stress that triggers adaptive responses, making cells more resilient over time .
How to Assess Oxidative Stress: Biomarkers You Should Know
Here’s where preventive healthcare gets exciting. We can’t directly measure free radicals—they’re too short-lived, lasting only fractions of a second . But we can measure the damage they leave behind, much like investigators studying footprints at a crime scene.
Types of Oxidative Stress Biomarkers
Scientists assess oxidative stress by measuring three main types of damage markers :
| Biomarker Type | What It Measures | Examples |
|---|---|---|
| DNA damage | Oxidized DNA excreted in urine | 8-hydroxy-2′-deoxyguanosine (8-OHdG) |
| Lipid peroxidation | Damaged fats from cell membranes | 8-isoprostaglandin-F2α (8-isoPGF2α), malondialdehyde (MDA) |
| Protein oxidation | Oxidized proteins | Protein carbonyls |
| Antioxidant levels | Body’s defense capacity | Glutathione, superoxide dismutase (SOD), catalase |
Advanced Testing Methods
Recent technological advances now allow simultaneous measurement of multiple biomarkers from a single urine sample. One validated method can quantify four key markers in just 12 minutes, detecting levels as low as 0.12 nanograms per milliliter .
Urinary biomarkers commonly measured include :
- 8-OHdG: DNA damage marker
- 8-isoPGF2α: Lipid peroxidation marker
- aMT6s: Melatonin metabolite (antioxidant-related)
- 11-DH-TXB2: Inflammation-related marker
Some comprehensive profiles now measure up to 16 markers of oxidative damage plus 32 genetic variants that affect how well your body produces antioxidant enzymes .
What About Genetics?
Your genes influence your antioxidant defenses. Key enzymes include :
- Superoxide dismutase (SOD): Converts superoxide radicals to hydrogen peroxide
- Catalase (CAT): Breaks down hydrogen peroxide into water
- Glutathione peroxidase (GPx): Neutralizes various peroxides
Genetic variations in these enzymes can affect your natural protection against oxidative stress—and knowing your status allows for personalized interventions .

Practical Ways to Reduce Oxidative Stress
The good news? You have significant control over your oxidative stress levels through lifestyle choices.
Dietary Strategies
Antioxidant-rich foods are your first line of defense :
| Food Category | Examples |
|---|---|
| Berries | Blueberries, strawberries, raspberries |
| Fruits | Cherries, citrus fruits, prunes, tomatoes, olives |
| Vegetables | Dark leafy greens, broccoli, carrots |
| Other | Nuts, seeds, green tea, coffee, dark chocolate |
Polyphenols, found abundantly in plant foods, are particularly effective at reducing oxidative damage markers and increasing antioxidant levels . The Mediterranean diet, rich in olive-derived polyphenols like oleuropein and hydroxytyrosol, has demonstrated geroprotective effects by activating key antioxidant pathways .
Lifestyle Modifications
Simple daily habits make a significant difference :
- Exercise moderately and regularly: This strengthens your body’s antioxidant defenses through hormesis
- Don’t smoke: Avoid both active and secondhand smoke
- Protect your skin: Use sunscreen to prevent UV-induced oxidative damage
- Limit alcohol: Excessive intake increases free radical production
- Prioritize sleep: Critical for maintaining antioxidant balance
- Reduce toxin exposure: Be mindful of pesticides, pollution, and industrial chemicals
Targeted Supplementation
For some individuals, personalized antioxidant supplements may be beneficial. These can include :
- Vitamins C and E
- Selenium
- Coenzyme Q10
- Melatonin
- Specialized polyphenol formulations
The key is personalization—matching supplements to your specific oxidative stress profile and genetic predispositions .
Oxidative stress is not just an abstract biological concept—it’s a measurable, manageable factor in how well we age. By understanding this balance between free radicals and antioxidants, we can take proactive steps to protect our cellular health.
The emerging field of oxidative stress assessment offers something unprecedented: the ability to detect aging processes before they manifest as disease and to tailor interventions specifically to each person’s needs .
Key takeaways:
- Oxidative stress is an imbalance between free radicals and antioxidants that accelerates aging
- It contributes to major chronic diseases including heart disease, diabetes, and neurodegeneration
- Biomarker testing can assess your personal oxidative stress levels through urine samples
- Lifestyle choices—diet, exercise, sleep, and avoiding toxins—powerfully influence your oxidative balance
- Personalized interventions based on testing and genetics offer the most effective approach
As research continues to evolve, one thing becomes clear: managing oxidative stress is not about eliminating it entirely—that’s neither possible nor desirable. Instead, the goal is balance, resilience, and informed choices that support healthy aging from the inside out.
115 Fundamental Questions About Hormesis
The science of controlled stress that builds strength, resilience & longevity
Click any question to reveal the answer
Fundamental Questions About Hormesis
1. What is hormesis?
Hormesis is the biological phenomenon where a low or moderate dose of a stressor produces a beneficial adaptive response, while a high dose is harmful. It’s the science behind “what doesn’t kill you makes you stronger.”
2. What does “The Science of Hormesis That Makes You Stronger” actually mean?
It refers to how mild, controlled stressors (exercise, fasting, heat, phytochemicals) activate repair pathways, antioxidant defenses, mitochondrial biogenesis and autophagy — ultimately making cells, tissues and the whole body more resilient and healthier.
3. How does hormesis work in the human body?
Mild stress signals danger → cells activate protective genes (Nrf2, AMPK, sirtuins, FOXO) → increased antioxidants, DNA repair, autophagy, mitochondrial renewal → net improvement in function and stress resistance.
4. What is a hormetic stressor?
A controlled challenge that is beneficial in small amounts: exercise, intermittent fasting, cold exposure, sauna, UV in moderation, certain phytochemicals (sulforaphane, curcumin, resveratrol), mild caloric restriction, etc.
5. Is hormesis scientifically proven?
Yes. Thousands of peer-reviewed studies since the 1940s (Calabrese et al.), widely accepted in toxicology, exercise physiology, nutrition science and aging research.
6. What is the hormetic dose-response curve?
A J-shaped or U-shaped curve: very low dose = no effect, optimal low-moderate dose = stimulation & benefit, high dose = toxicity/inhibition.
7. Why is hormesis described as biphasic?
Because it has two opposing phases: beneficial stimulation at low dose, inhibition or toxicity at high dose — the classic biphasic dose-response.
8. What is the difference between stress and hormetic stress?
Normal/chronic stress overwhelms repair capacity and causes net damage. Hormetic stress is brief, mild and recoverable — the body overcompensates and becomes stronger.
9. Can stress really be good for you?
Yes — when it’s the right type (hormetic), dose and duration. That’s why moderate exercise, fasting and sauna improve health markers while chronic stress accelerates aging.
10. Is hormesis the same as resilience?
No — hormesis is the process (repeated mild stress → adaptation). Resilience is the outcome — increased capacity to handle future stress.
🔬 Cellular & Biological Mechanisms
11. What happens at the cellular level during hormesis?
Mild damage → activation of transcription factors → upregulation of protective genes → mitochondrial biogenesis, antioxidant enzymes, autophagy, DNA repair → stronger, cleaner cells.
12. How does hormesis activate antioxidant defenses?
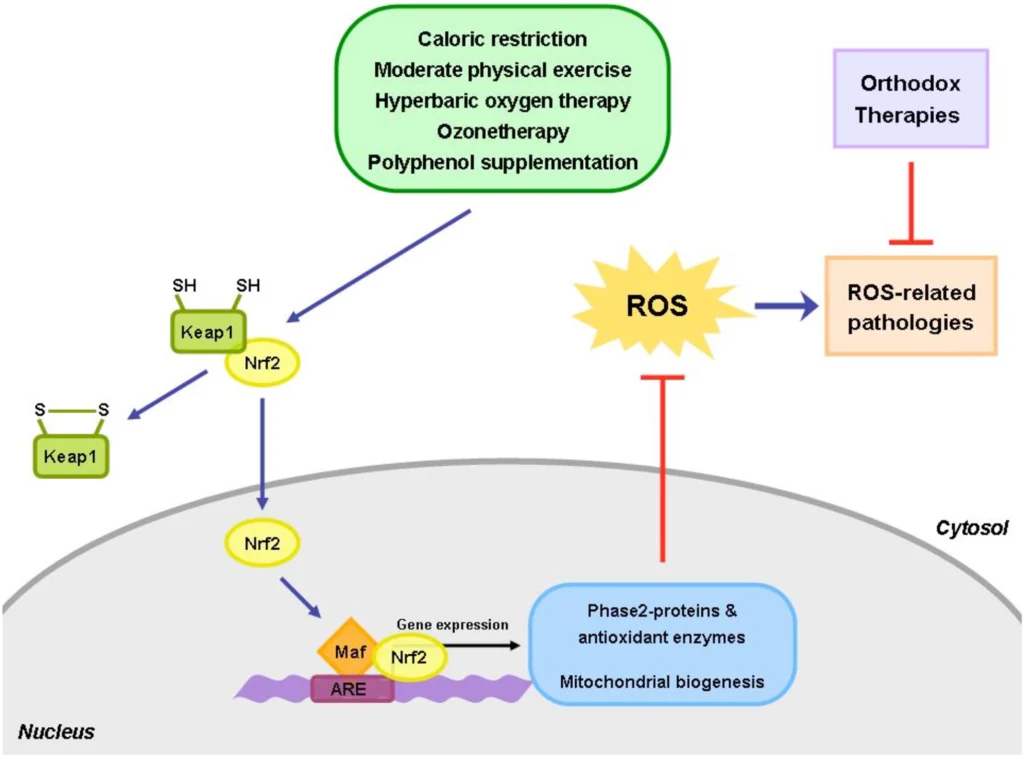
It mildly increases ROS → Nrf2 translocation to nucleus → transcription of SOD, catalase, glutathione peroxidase, HO-1 and many other protective enzymes.
13. What is the role of Nrf2 in hormesis?
Nrf2 is the master regulator of the antioxidant response. Hormetic stressors dissociate Nrf2 from Keap1 → Nrf2 enters nucleus → turns on ~200–500 cytoprotective genes.
14. How does AMPK contribute to adaptive stress responses?
AMPK senses low energy → inhibits mTOR, activates autophagy, fat oxidation, mitochondrial biogenesis and antioxidant defenses — key mediator of fasting & exercise benefits.
15. Does hormesis increase mitochondrial function?
Yes — through biogenesis (more mitochondria), improved efficiency (better ETC), and selective mitophagy (removal of damaged ones).
16. What is mitochondrial biogenesis?
The creation of new mitochondria, mainly driven by PGC-1α. Triggered by exercise, cold, fasting, resveratrol, etc. → more energy, less ROS leakage.
17. How does hormesis stimulate autophagy?
Mild stress → AMPK activation + mTOR inhibition → ULK1 → autophagosome formation → recycling of damaged proteins/organelles → cellular rejuvenation.
18. What are heat shock proteins and why do they matter?
HSPs (HSP70, HSP90, HSP27…) are molecular chaperones. They refold misfolded proteins, prevent aggregation and protect during stress. Upregulated by heat, exercise, phytochemicals.
19. How does hormesis affect inflammation?
Low-dose stress reduces chronic (NF-κB driven) inflammation while acutely increasing beneficial inflammatory signals that resolve quickly.
20. Does hormesis influence gene expression?
Yes — epigenetically and transcriptionally. It activates sirtuins, Nrf2, FOXO, HSF1 → changes expression of thousands of genes toward repair and longevity.
21. Can hormesis slow aging?
Strong evidence from model organisms and humans: hormetic interventions (CR, exercise, heat/cold) consistently extend healthspan and often lifespan via shared pathways.
22. How does oxidative stress relate to hormesis?
Low-level ROS acts as a signaling molecule that triggers adaptive antioxidant responses. Too much ROS = damage. Hormesis exploits the beneficial signaling zone.
23. Is hormesis linked to longevity pathways?
Yes — it overlaps heavily with caloric restriction, sirtuins, mTOR inhibition, AMPK, insulin/IGF-1 signaling — core longevity pathways.
24. How does hormesis improve metabolic flexibility?
It trains cells to switch efficiently between glucose and fat oxidation (via AMPK, PGC-1α, mitophagy) → better insulin sensitivity and energy homeostasis.
25. Does hormesis strengthen the immune system?
Yes — mild stress enhances innate immunity, improves surveillance, reduces inflammaging, and can increase vaccine response in older adults.
🏋️ Exercise & Physical Training
26. Why is exercise considered a hormetic stressor?
It causes controlled muscle damage, ROS production, energy depletion → triggers repair, mitochondrial biogenesis, antioxidant upregulation and anti-inflammatory effects.
27. How much exercise is optimal for hormesis?
Most studies show 150–300 min moderate or 75–150 min vigorous per week. Individual sweet spot varies — monitor HRV/recovery to avoid overtraining.
28. Can overtraining cancel hormetic benefits?
Yes — chronic overtraining causes sustained inflammation, cortisol elevation, immunosuppression and oxidative damage — tipping past the hormetic zone.
29. Is high-intensity interval training hormetic?
Yes — HIIT is one of the strongest hormetic protocols: brief intense effort → large AMPK/Nrf2 activation → excellent mitochondrial and metabolic adaptations.
30. Does strength training follow hormesis principles?
Yes — progressive overload creates repeated mild damage → supercompensation → increased muscle mass, strength, bone density and metabolic health.
31. How does progressive overload relate to hormesis?
It’s textbook hormesis: gradually increasing the stressor (weight, reps, volume) keeps the body in the adaptive zone without crossing into chronic damage.
32. Is endurance training hormetic?
Yes — moderate endurance increases mitochondrial density, capillary networks, fat oxidation and antioxidant capacity — classic hormetic adaptation.
33. Can beginners apply hormesis safely in workouts?
Yes — start very low (walking, bodyweight, light resistance) and progress slowly. The principle works at any level — dose is relative to current fitness.
34. How do rest days influence hormetic adaptation?
Essential — adaptation occurs during recovery. Rest allows supercompensation (muscle repair, mitochondrial renewal, antioxidant upregulation).
35. Does soreness mean hormesis is working?
Not necessarily. Mild DOMS can indicate adaptation, but severe or prolonged soreness usually means you exceeded the hormetic zone.
⏳ Fasting & Nutrition
36. Is intermittent fasting an example of hormesis?
Yes — one of the most studied. Short energy deficit → AMPK/sirtuins/autophagy → metabolic flexibility, reduced inflammation, cellular cleanup.
37. How long should you fast for hormetic benefits?
Most benefits appear between 12–24 hours (16:8 common). Autophagy ramps up ~16–18 h, deeper effects ~24–36 h. Longer needs medical supervision.
38. Does caloric restriction extend lifespan via hormesis?
Yes — in many species. 20–40% CR activates sirtuins, AMPK, reduces mTOR/IGF-1 → hormetic stress response linked to longevity.
39. What is the relationship between autophagy and fasting?
Fasting strongly induces autophagy via AMPK/mTOR inhibition → cellular recycling peaks after ~16–24 hours of fasting.
40. Can fasting be harmful if overdone?
Yes — prolonged fasting without preparation can cause muscle loss, electrolyte imbalance, gallstones, weakened immunity in some people.
41. Is time-restricted eating hormetic?
Yes — eating within 8–12 hour window creates daily mild energy stress → improves circadian rhythm, insulin sensitivity and gut repair.
42. Does protein restriction stimulate hormesis?
Yes — moderate protein restriction (especially BCAAs) lowers mTOR/IGF-1 → activates autophagy and longevity pathways similar to CR.
43. Can certain plant compounds trigger hormesis?
Yes — “xenohormesis”: low-dose phytochemicals (sulforaphane, curcumin, resveratrol, quercetin) act as mild stressors → Nrf2 activation.
44. What are hormetic phytochemicals?
Plant defense molecules that in tiny amounts stress human cells beneficially: sulforaphane (broccoli sprouts), curcumin, resveratrol, quercetin, catechins, allicin.
45. Is coffee a hormetic stressor?
Yes — moderate coffee (caffeine + polyphenols) activates Nrf2, AMPK, increases antioxidant enzymes and shows U-shaped longevity association.
❄️🔥 Temperature Exposure
46. Is cold exposure a form of hormesis?
Yes — brief cold stress → norepinephrine, brown fat activation, mitochondrial biogenesis, improved insulin sensitivity.
47. How does cold plunging activate resilience pathways?
Cold → sympathetic surge → PGC-1α → mitochondrial biogenesis in muscle & fat; also increases adiponectin and reduces inflammation.
48. Are ice baths safe for everyone?
No — caution in cardiovascular disease, Raynaud’s, uncontrolled hypertension, pregnancy. Start short (30–60 s) and warm up slowly.
49. Does sauna use follow hormesis principles?
Yes — heat stress → HSF1 → heat shock proteins, Nrf2, improved cardiovascular function, reduced all-cause mortality (Finnish studies).
50. How long should sauna sessions last for hormetic effects?
15–25 minutes at 80–100°C (176–212°F), 2–4×/week. Benefits plateau and risk rises beyond ~30 min per session.
51. Can alternating hot and cold amplify benefits?
Yes — contrast therapy (sauna → cold plunge) creates larger hormetic signal, improves circulation, reduces inflammation more than single modality.
52. What is the cardiovascular impact of thermal stress?
Sauna mimics moderate cardio (↑ heart rate, vasodilation); cold causes vasoconstriction then rebound. Both improve endothelial function long-term.
53. Can too much cold exposure be harmful?
Yes — prolonged/excessive cold → hypothermia, immunosuppression, cardiovascular strain in susceptible people.
54. How often should you practice thermal hormesis?
2–5×/week (sauna or cold) is common in studies showing benefits without diminishing returns.
55. Does climate adaptation reflect hormesis?
Yes — repeated exposure to heat/cold → physiological adaptations (sweating efficiency, brown fat, vascular tone) — classic hormesis.
🧬 Longevity & Aging
56. Does hormesis increase lifespan?
In many model organisms yes. In humans — strong evidence for healthspan extension; lifespan data still emerging but supportive.
57. Can hormesis delay biological aging?
Yes — reduces epigenetic age acceleration, preserves telomere function, lowers inflammation, improves mitochondrial health — measurable on biological clocks.
58. What is the link between hormesis and longevity research?
Hormesis overlaps almost completely with CR mimetics, sirtuin activators, mTOR inhibitors — core interventions studied for longevity.
59. Do centenarians show signs of hormetic adaptation?
Many do — lifelong moderate stressors (physical labor, fasting periods, phytochemical-rich diets) correlate with exceptional longevity.
60. Is hormesis part of anti-aging strategies?
Yes — one of the few evidence-based lifestyle levers (exercise, fasting, heat/cold, xenohormetics) that reliably slow aging markers.
61. Does hormesis reduce cellular senescence?
Yes — by enhancing autophagy, reducing chronic inflammation and oxidative damage — fewer senescent cells accumulate.
62. How does hormesis affect telomeres?
Mild stress can slow telomere shortening via telomerase upregulation and reduced oxidative/inflammatory burden on telomeres.
63. Can hormesis prevent chronic disease?
Yes — reduces risk factors for diabetes, CVD, neurodegeneration, cancer via improved metabolic health, lower inflammation, better DNA stability.
64. Is oxidative stress always harmful?
No — low-level ROS is a vital signaling molecule. Hormesis exploits this signaling while preventing excess damage.
65. Does mild stress enhance repair mechanisms long term?
Yes — repeated hormetic exposure upregulates baseline DNA repair enzymes, antioxidant capacity and chaperone levels.
🧘 Psychological & Mental Resilience
66. Can psychological stress be hormetic?
Yes — controlled, short-term psychological challenges (public speaking, cold showers, voluntary discomfort) build mental toughness.
67. Is mental resilience built through hormesis?
Yes — repeated exposure to manageable stress → habituation, reduced amygdala reactivity, stronger prefrontal control.
68. Does exposure therapy use hormetic principles?
Yes — gradual, controlled exposure to feared stimuli → habituation and reduced fear response — classic psychological hormesis.
69. Can discomfort improve emotional strength?
Yes — voluntary discomfort trains tolerance to negative emotions → lower reactivity, greater emotional regulation.
70. How does hormesis relate to grit?
Grit = perseverance through difficulty. Hormetic practice trains exactly that — seeing stress as growth opportunity.
71. Is social stress ever beneficial?
Yes — moderate social challenges (networking, difficult conversations) → improved social skills, confidence, emotional intelligence.
72. Can controlled adversity improve confidence?
Yes — overcoming small, chosen adversities creates mastery experiences → higher self-efficacy and confidence.
73. How does stress inoculation work?
Gradual exposure to stressors in training → builds coping skills and physiological habituation → better performance under real stress.
74. Does hormesis influence neuroplasticity?
Yes — mild stress upregulates BDNF, enhances synaptic plasticity, promotes new neural connections — especially in hippocampus.
75. Can hormesis protect against burnout?
Yes — by improving stress tolerance and recovery capacity, regular hormetic practice reduces risk of burnout from chronic overload.
⚖️ Safety & Risk Management
76. How do you find your optimal stress dose?
Start low → monitor subjective (energy, mood) + objective (HRV, sleep, recovery) markers → gradually increase until benefits plateau or side effects appear.
77. What are signs of excessive stress?
Poor sleep, persistent fatigue, mood swings, stalled progress, frequent illness, elevated resting HR, low HRV, chronic soreness.
78. Who should avoid hormetic stressors?
Pregnant women, acute illness, severe cardiovascular disease, eating disorders, uncontrolled hypertension, adrenal fatigue — consult doctor.
79. Can people with chronic illness apply hormesis?
Often yes — but very carefully and under supervision (e.g., gentle movement, short fasts, mild heat). Individualization is critical.
80. Is hormesis safe for older adults?
Yes — and often more important. Start very gently; benefits for muscle preservation, cognition, cardiovascular health are well documented.
81. How do you recover properly after stress exposure?
Prioritize sleep (7–9 h), nutrition (protein + antioxidants), hydration, active recovery (walking), stress management (breathing/meditation).
82. Can hormesis weaken the body if misapplied?
Yes — too frequent, too intense, or without recovery → chronic stress state, immunosuppression, injury, burnout.
83. How does sleep affect hormetic benefits?
Critical — most adaptation (muscle repair, mitochondrial renewal, memory consolidation) occurs during deep sleep. Poor sleep blunts hormesis.
84. Can combining stressors be dangerous?
Yes — stacking multiple intense stressors (e.g., hard workout + long fast + ice bath same day) can easily exceed recovery capacity.
85. What medical conditions require caution?
Heart disease, arrhythmias, Raynaud’s, thyroid disorders, autoimmune flares, eating disorders, pregnancy, recent surgery — always consult physician.
🧪 Advanced & Controversial Topics
86. Is radiation hormetic?
Low-dose ionizing radiation shows hormetic effects in some studies (DNA repair upregulation, cancer reduction), but high-dose is clearly carcinogenic — highly debated.
87. Are toxins ever beneficial in small doses?
Yes — many hormetic agents are mild toxins (xenobiotics): alcohol (low dose), heavy metals (trace), phytochemicals. Dose makes the poison.
88. Is alcohol hormetic?
Possibly in very low doses (1 drink/day) — mild Nrf2 activation, cardiovascular benefits in some populations. Risk rapidly outweighs benefit above that.
89. Can environmental pollutants trigger adaptive responses?
Some low-level pollutants show hormetic effects in lab studies, but real-world chronic exposure usually causes net harm.
90. Is hormesis accepted by all scientists?
Very widely accepted in toxicology and physiology. Some debate remains on low-dose radiation and certain xenobiotics.
91. What are criticisms of the hormesis theory?
Over-extrapolation to radiation/toxins, difficulty defining “low dose” in humans, potential publication bias, individual variability.
92. Does hormesis apply to pharmaceuticals?
Yes — many drugs show hormetic dose-responses (e.g., aspirin, statins, metformin, rapamycin at low dose).
93. Can microdosing be considered hormetic?
Possibly — very low doses of psychedelics may induce mild neuroplasticity/stress responses without full effects — emerging research area.
94. Is oxidative stress supplementation counterproductive?
High-dose isolated antioxidants can blunt hormetic signaling (ROS needed for adaptation). Food-based antioxidants usually better.
95. Does antioxidant overuse blunt hormetic benefits?
Yes — mega-dose vitamin C/E can reduce exercise-induced adaptations (mitochondrial biogenesis, insulin sensitivity) by neutralizing signaling ROS.
🧭 Practical Application & Strategy
96. How do you build a hormesis routine?
Start with 1–2 mild stressors (e.g., 16:8 fasting + daily walk), add slowly, monitor recovery, cycle intensity, prioritize sleep/nutrition.
97. How often should hormetic stressors be applied?
2–6×/week depending on type (daily movement, 3–5× exercise, 2–4× sauna/cold, 5–7× time-restricted eating). Recovery is key.
98. Should stressors be cycled?
Yes — prevents adaptation plateau. Alternate modalities (e.g., HIIT one month, endurance next) or vary intensity.
99. Can you combine fasting and exercise safely?
Yes — fasted training often enhances fat adaptation. Avoid very intense sessions when fasted >18–20 h if new to it.
100. What is the minimum effective hormetic dose?
Varies — 10–15 min brisk walk, 12–14 h overnight fast, 1–2 min cold shower, handful broccoli sprouts can all initiate mild benefits.
101. How do you measure adaptation?
Subjective: energy, mood, recovery speed. Objective: HRV, resting HR, fasting glucose, inflammation markers, strength gains, sleep quality.
102. Are wearable devices useful for monitoring stress?
Yes — track HRV (recovery), resting HR (overtraining), sleep stages, activity levels — guide dosing and recovery.
103. Can HRV track hormetic response?
Yes — acute drop after stressor → rise during recovery = good adaptation. Chronic low HRV = overdoing it.
104. How long does adaptation take?
Acute effects: hours–days. Structural changes (mitochondria, muscle): 4–12 weeks. Long-term resilience: months–years of consistency.
105. How do you periodize hormetic stress?
Use cycles: 4–8 weeks build → 1 week deload, or seasonal focus (summer heat, winter cold), or undulating weekly intensity.
🌍 Broader Conceptual Questions
106. Is comfort culture reducing resilience?
Many argue yes — constant comfort removes hormetic signals → lower stress tolerance, weaker physiological/psychological adaptations.
107. Did humans evolve with hormesis?
Yes — intermittent fasting, temperature swings, physical exertion, toxin exposure from plants — all shaped our stress-response systems.
108. Is modern life too stress-free biologically?
Physically yes for many — no famine, constant temperature, low physical demand — potentially reducing hormetic “training” signals.
109. Can hormesis explain why challenges build character?
Yes — overcoming manageable adversity → neuroplasticity, grit, self-efficacy, emotional regulation — biological basis of character growth.
110. Is hormesis the foundation of strength training?
Yes — progressive overload is applied hormesis: repeated mild damage → supercompensation → stronger tissue.
111. Does hormesis apply beyond biology?
Yes — analogous in psychology (exposure therapy), economics (antifragility – Taleb), learning (desirable difficulties), business (controlled risk).
112. Can hormesis principles improve productivity?
Yes — deliberate short bursts of deep work, scheduled discomfort, fasting windows → better focus, energy management, resilience to pressure.
113. Is hormesis the science behind “what doesn’t kill you makes you stronger”?
Exactly — Nietzsche’s phrase captures the essence of hormesis: survivable stress → adaptation and growth.
114. How does hormesis differ from toxic stress?
Hormesis = recoverable, adaptive stress. Toxic stress = overwhelming, chronic, damaging (ACEs, prolonged trauma, burnout).
115. Is hormesis the key to sustainable strength?
One of the most important keys — sustainable strength (physical, mental, metabolic) comes from repeated, recoverable challenges that drive adaptation without breakdown.
Ready to apply hormesis intelligently?
Measure real progress with oxidative stress & DNA repair biomarkers.
Explore DNA Repair & Oxidative Stress Testing →Get Free Personalized Hormesis Recommendations

Dr. Mohammed Abdul Azeem Siddiqui, MBBS, M.Tech (Biomedical Engineering – VIT, Vellore)
Registered Medical Practitioner – Reg. No. 39739
Physician • Clinical Engineer • Preventive Diagnostics Specialist
Dr. Mohammed Abdul Azeem Siddiqui is a physician–engineer with over 30 years of dedicated clinical and biomedical engineering experience, committed to transforming modern healthcare from late-stage disease treatment to early detection, preventive intelligence, and affordable medical care.
He holds an MBBS degree in Medicine and an M.Tech in Biomedical Engineering from VIT University, Vellore, equipping him with rare dual expertise in clinical medicine, laboratory diagnostics, and medical device engineering. This allows him to translate complex laboratory data into precise, actionable preventive strategies.
Clinical Mission
Dr. Siddiqui’s professional mission centers on three core pillars:
Early Disease Detection
Identifying hidden biomarker abnormalities that signal chronic disease years before symptoms appear — reducing complications, hospitalizations, and long-term disability.
Preventive Healthcare
Guiding individuals and families toward longer, healthier lives through structured screenings, lifestyle intervention frameworks, and predictive diagnostic interpretation.
Affordable Evidence-Based Treatment
Delivering cost-effective, scientifically validated care accessible to people from all socioeconomic backgrounds.
Clinical & Technical Expertise
Across three decades of continuous practice, Dr. Siddiqui has worked extensively with:
Advanced laboratory analyzers and automation platforms
• Cardiac, metabolic, renal, hepatic, endocrine, and inflammatory biomarker systems
• Preventive screening and early organ damage detection frameworks
• Clinical escalation pathways and diagnostic decision-support models
• Medical device validation, calibration, compliance, and patient safety standards
He is recognized for identifying subclinical biomarker shifts that predict cardiovascular disease, diabetes, fatty liver, kidney disease, autoimmune inflammation, neurodegeneration, and accelerated biological aging long before conventional diagnosis.
Role at IntelliNewz
At IntelliNewz, Dr. Siddiqui serves as Founder, Chief Medical Editor, and Lead Clinical Validator. Every article published is:
Evidence-based
• Clinically verified
• Technology-grounded
• Free from commercial bias
• Designed for real-world patient and physician decision-making
Through his writing, Dr. Siddiqui shares practical health intelligence, early warning signs, and preventive strategies that readers can trust — grounded in decades of frontline medical practice.
Contact:
powerofprevention@outlook.com
📌 Disclaimer: The content on IntelliNewz is intended for educational purposes only and does not replace personalized medical consultation. For individual health concerns, please consult your physician.