The Next Wave of CSF Biomarkers—From Synapses to Exosomes
The Future is Molecular
These seven biomarkers represent a shift from viewing the CSF as a static fluid to a dynamic repository of brain health. They are moving us toward a future where we can detect the “whisper” of disease—be it synaptic loss, cryptic exon mis-splicing, or smoldering inflammation—long before it becomes a clinical “scream.” While most of these tests remain in the research domain, their translation to the clinic will likely define the next decade of neurology, turning currently untreatable or unpreventable conditions into manageable ones.
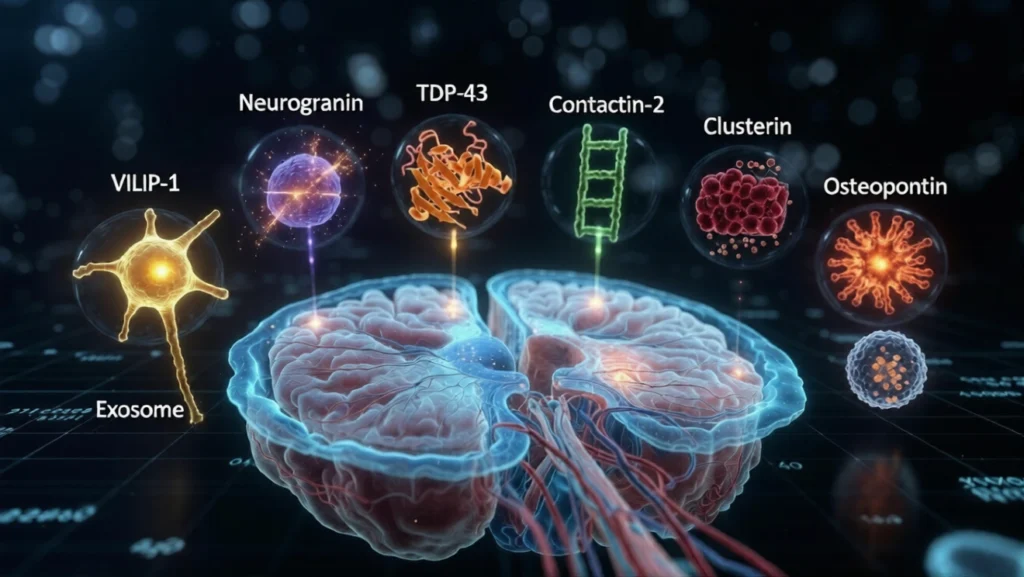
1. Visinin-Like Protein-1 (VILIP-1)2. Neurogranin A postsynaptic protein involved in synaptic plasticity.3. TAR DNA-Binding Protein-43 (TDP-43) Abnormal aggregation of this protein is a hallmark of several neurodegenerative disorders. Associated diseases: Amyotrophic lateral sclerosis Frontotemporal dementia. Contactin-2 (TAG-1) A neuronal adhesion molecule involved in axon guidance and synaptic integrity. Research significance: Reduced CSF levels correlate with axonal degeneration. Associated diseases: Multiple sclerosis Alzheimer’s disease5. Clusterin (Apolipoprotein J)6. Osteopontin A pro-inflammatory cytokine-like protein involved in immune signaling. Clinical significance:7. Extracellular Vesicle (Exosome) Biomarkers CSF neuronal exosomes carry proteins, lipids, and RNA molecules that mirror brain pathology. Important cargo molecules: phosphorylated tau α-synuclein microRNAs what are these
Here is a detailed breakdown of the seven additional biomarkers you listed. While the previous article focused on emerging and highly specific proteins (like HDGFL2), these seven markers represent a mix of synaptic proteins, structural proteins, and inflammatory mediators that are currently being validated for their predictive utility.
1. Visinin-Like Protein-1 (VILIP-1)
- What it is: A neuronal calcium sensor protein primarily located in the cell bodies and dendrites of neurons. It is involved in regulating synaptic transmission and plasticity via calcium signaling pathways.
- Pathophysiology: When neurons are injured or undergo excitotoxicity (overstimulation by neurotransmitters like glutamate), calcium homeostasis fails, leading to the release of VILIP-1 into the interstitial fluid and ultimately the CSF.
- Clinical Significance:
- Alzheimer’s Disease (AD): VILIP-1 is considered a marker of neuronal injury, similar to total tau (t-tau), but it may be more specific to cortical neurons. Elevated CSF VILIP-1 correlates strongly with cognitive decline and brain atrophy.
- Predictive Value: It can differentiate stable Mild Cognitive Impairment (MCI) from MCI that will progress to AD dementia. It often rises in tandem with tau, providing a combined measure of injury and pathology.
2. Neurogranin (Ng)
- What it is: A postsynaptic protein expressed predominantly in the dendrites of neurons in the cortex and hippocampus. It plays a critical role in long-term potentiation (LTP)—the molecular basis of memory and learning—by regulating calmodulin.
- Pathophysiology: Loss of synaptic function is the strongest correlate of cognitive decline. Neurogranin leaks into the CSF specifically when postsynaptic elements are degenerating.
- Clinical Significance:
- Specificity for AD: Unlike some generic neuronal injury markers, elevated CSF Neurogranin appears to be relatively specific to Alzheimer’s disease compared to other neurodegenerative diseases like Parkinson’s or FTD.
- Early Detection: It is elevated in prodromal AD (MCI due to AD) and predicts future cognitive decline and hippocampal atrophy.
3. TAR DNA-Binding Protein-43 (TDP-43)
- What it is: A nuclear protein that regulates gene expression and RNA processing. Under normal conditions, it resides in the nucleus. In disease, it mislocalizes to the cytoplasm, aggregates, and becomes hyperphosphorylated.
- Pathophysiology: TDP-43 pathology is the hallmark of ALS and a subtype of Frontotemporal dementia (FTD) . It is also present in a significant percentage of Alzheimer’s disease patients (called LATE: Limbic-predominant Age-related TDP-43 Encephalopathy), where it accelerates cognitive decline beyond what amyloid and tau alone would cause.
- Clinical Significance & Challenges:
- CSF Utility: Measuring total TDP-43 in CSF has been challenging because it is also present in blood cells and can be contaminated by cell lysis. However, new assays targeting *phosphorylated TDP-43* (p-TDP-43) or specific N-terminal fragments are being developed to specifically detect the pathological form, aiming to predict TDP-43 pathology in vivo (currently only possible at autopsy).
4. Contactin-2 (TAG-1)
- What it is: A cell adhesion molecule (part of the Contactin family) expressed on neurons. It is anchored to the cell membrane and is involved in axon guidance, neuronal migration, and maintaining the integrity of the nodes of Ranvier (crucial for rapid nerve conduction).
- Pathophysiology: Levels of Contactin-2 decrease in the CSF when there is significant axonal damage or disruption of the neuronal membrane.
- Clinical Significance:
- Multiple Sclerosis (MS): Reduced CSF levels correlate with axonal degeneration in progressive MS.
- Alzheimer’s Disease: It is being investigated as part of a panel of synaptic proteins (along with Neurogranin and SNAP-25) to understand the cascade of synaptic and axonal breakdown.
5. Clusterin (Apolipoprotein J)
- What it is: A multifunctional glycoprotein and a major chaperone (protein “quality control” agent) in the brain. It is closely related to Apolipoprotein E (ApoE).
- Pathophysiology: Clusterin is involved in lipid transport, complement regulation (inflammation), and clearing extracellular misfolded proteins. When the brain is stressed by protein aggregation, astrocytes and neurons upregulate Clusterin.
- Clinical Significance:
- Alzheimer’s Disease: Clusterin binds to soluble Aβ, influencing its clearance and aggregation. Elevated CSF Clusterin is associated with faster cognitive decline and brain atrophy in AD.
- Genetic Risk: Variants in the CLU gene (which encodes Clusterin) are a major genetic risk factor for late-onset Alzheimer’s disease.
- Dual Role: It serves as both a marker of proteotoxic stress and a potential therapeutic target.
6. Osteopontin (OPN)
- What it is: A pro-inflammatory cytokine and matricellular protein. In the CNS, it is primarily produced by microglia (the brain’s resident immune cells) and macrophages.
- Pathophysiology: OPN promotes the migration and activation of immune cells. In neuroinflammatory conditions, it drives the chronic, “smoldering” inflammation that standard anti-inflammatory drugs often miss.
- Clinical Significance:
- Multiple Sclerosis: CSF Osteopontin levels correlate with disability progression and the conversion from Relapsing-Remitting MS to Secondary Progressive MS. It is a marker of the compartmentalized inflammation behind the blood-brain barrier that drives progression.
- ALS: High levels are associated with faster disease progression.
7. Extracellular Vesicle (Exosome) Biomarkers
- What they are: Exosomes are tiny nanoparticles (30-150nm) released by all cells, including neurons, astrocytes, and oligodendrocytes. They are essentially “message in a bottle” packages containing proteins, lipids, mRNA, and microRNA (miRNA) that reflect the state of their parent cell.
- Why they are revolutionary: CSF is a complex fluid containing soluble proteins. Exosome analysis allows researchers to “enrich” for neuronal material.
- Isolation: Techniques allow the immunocapture of exosomes derived specifically from neurons (using L1CAM antibody) or astrocytes (using GLAST antibody).
- Important Cargo Molecules:
- Phosphorylated Tau (p-tau): Found in neuronal exosomes, it may predict the spread of tau pathology years before cortical involvement.
- α-Synuclein: In Parkinson’s disease, exosomal α-synuclein in CSF may distinguish PD from other synucleinopathies (like Multiple System Atrophy) and detect pathology in the prodromal phase.
- microRNAs (miRNAs): These small non-coding RNAs regulate gene expression. Specific miRNA signatures in CSF exosomes are being investigated as “fingerprints” for early-stage disease, particularly in ALS and Huntington’s disease, where they can reflect neuronal stress before cell death.
The human cerebrospinal fluid (CSF) is more than just a protective cushion for the brain; it is a dynamic window into the molecular health of the central nervous system. For years, clinical neurology has relied on a handful of core biomarkers—such as amyloid-beta, total tau, and neurofilament light chain—to confirm the presence of diseases like Alzheimer’s or multiple sclerosis. However, these markers often register significant changes only after substantial, and often irreversible, neuronal damage has occurred.
To truly predict and preempt neurological disease, researchers are now delving deeper into the CSF to capture the subtle whispers of pathology that precede clinical silence. This has led to the investigation of a new class of markers that target specific sub-compartments of the neuron and the nuanced processes of early degeneration.
This article explores seven such emerging biomarkers currently at the forefront of advanced neurology and neuroimmunology research:
We begin with Visinin-Like Protein-1 (VILIP-1) and Neurogranin, two proteins that serve as precise barometers of neuronal integrity and synaptic function. Their elevation in CSF offers some of the earliest indications of the synaptic dysfunction that underlies cognitive decline.
Next, we examine TAR DNA-Binding Protein-43 (TDP-43) , a nuclear protein whose misdirection and aggregation defines the pathology of ALS and frontotemporal dementia, representing a critical target for detecting proteinopathy at its source.
We then turn to the structural and immune interface of the brain with Contactin-2 (TAG-1) , a neuronal adhesion molecule whose reduction signals axonal degradation, and Clusterin (Apolipoprotein J) , a versatile chaperone involved in both lipid transport and the clearance of toxic proteins.
The inflammatory landscape is captured by Osteopontin, a pro-inflammatory cytokine that acts as a sentinel for the chronic, “smoldering” inflammation driving progression in diseases like multiple sclerosis.
Finally, we look inside the smallest messengers of the brain: Extracellular Vesicles (Exosomes) . These nanoparticles, released by neurons and glia, carry cargo—including phosphorylated tau, α-synuclein, and microRNAs—that offers a concentrated, cell-specific snapshot of pathology, potentially years before clinical manifestations.
Together, these seven markers represent a shift from generalized neurodegeneration to patho-mechanism specific detection. They are the vanguard of a future where neurologists can diagnose, predict, and monitor disease with unprecedented molecular precision.