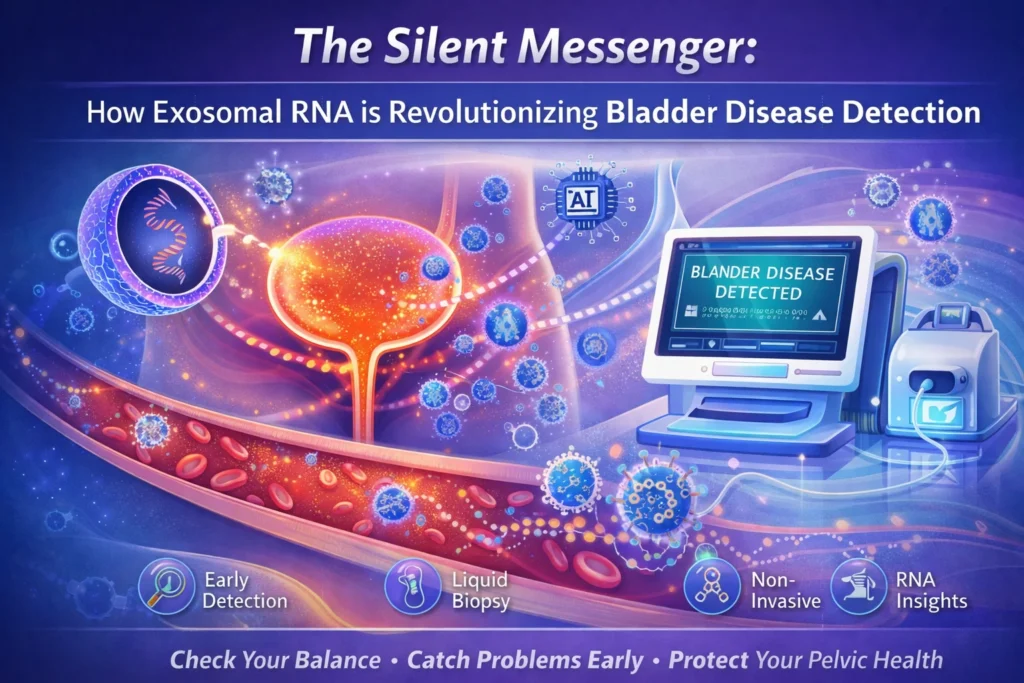
For decades, diagnosing bladder cancer has relied on a uncomfortable trade-off. The gold standard—cystoscopy—involves threading a camera into the bladder. It is invasive, expensive, and often dreaded by patients. The non-invasive alternative, urine cytology (looking for cancer cells under a microscope), is famously unreliable, missing up to 52% of cancers, especially in the early stages .
But hidden inside your urine is a treasure trove of genetic information that is changing everything. It comes in the form of microscopic bubbles called exosomes, and the RNA they carry is proving to be one of the most powerful “liquid biopsy” tools of the decade.
This is the story of exosomal RNA—the silent messenger that could replace the camera with a urine cup.
Part 1: What are Exosomes? (The Body’s “Memory Sticks”)
Imagine every cell in your body, including cancerous ones, sending out tiny, nano-sized shipping containers. These are exosomes. Ranging from 30 to 150 nanometers (thousands of times smaller than a human hair), these vesicles are pinched off from the cell membrane and travel throughout the body via bodily fluids like blood and urine .
Why are they so valuable to doctors? Because inside each exosome, the parent cell packs a biological “memory card.”
- Proteins
- mRNA (messenger RNA)
- miRNA (micro RNA)
- lncRNA (long non-coding RNA)
The magic of the exosome is that it has a protective lipid (fat) coating. In the harsh environment of urine, which is full of enzymes that destroy DNA and RNA, a free-floating piece of genetic material degrades instantly. But an exosome acts like a microscopic submarine, safely delivering its RNA cargo to be analyzed in a lab .
For bladder cancer detection, this is a game-changer. Because the bladder is a reservoir, cancerous cells in the bladder wall shed exosomes directly into the urine. You are literally “peeing out” the genetic signature of the tumor.
Part 2: Why Exosomal RNA Beats Traditional Tests
Current non-invasive tests look for blood, abnormal proteins, or loose cancer cells. Exosomal RNA analysis looks for the instructions the cancer cells are sending.
A landmark meta-analysis published in 2024 (reviewing 21 studies with 3,348 participants) found that urine-derived exosomes have a combined sensitivity of 75% and specificity of 77% for diagnosing bladder cancer, with an impressive diagnostic accuracy (AUC) of 0.83 .
To put that in perspective: This means a urine exosome test is significantly more reliable than looking for cells under a microscope (cytology) and is approaching the accuracy of invasive surgery, but without the pain or risk of infection.
The Three Types of Exosomal RNA (The “Detective Trio”)
Scientists are currently looking at three specific types of RNA inside these exosomes to catch disease:
- miRNA (Micro RNA): The regulator. These small molecules control gene expression. Specific miRNA signatures (like the let-7c cluster) have been found to be highly accurate in distinguishing cancer from healthy tissue using machine learning algorithms .
- lncRNA (Long Non-Coding RNA): The structural map. Recent breakthrough studies have identified specific lncRNA fingerprints (such as CCDC148-AS1) that are significantly over-expressed in non-muscle-invasive bladder cancer (NMIBC) patients, even in the very early stages .
- mRNA (Messenger RNA): The blueprint. Markers like KRT17 and MDK have been validated in multi-center prospective studies to monitor cancer recurrence over time .
Part 3: The Latest Breakthroughs (2024-2025)
The science is moving fast. Here is what has happened in the last 12 months that has urologists excited.
1. The “100% Accuracy” Study
In September 2025, a team of researchers published a study in the Chemical Engineering Journal unveiling a new imaging technique for urinary exosomes. Using engineered artificial vesicles that fuse specifically with cancer-derived exosomes, they were able to analyze five different miRNAs simultaneously. The result? Both sensitivity and specificity reached 100% in diagnosing bladder cancer .
While this is still in the research phase, it proves that a perfect urine test is scientifically possible.
2. Monitoring Recurrence Without Surgery
Bladder cancer has one of the highest recurrence rates of any cancer (up to 50% for high-risk NMIBC) . This means patients usually undergo invasive cystoscopy every 3 to 6 months. A 2024 prospective study tracked 189 patients over 19 months. They found that levels of the exosomal mRNA marker KRT17 were significantly higher in patients whose cancer returned, and it naturally dropped in those who remained cancer-free. This suggests that a simple urine test could soon tell you if you need another camera exam or if you are safe .
3. Machine Learning Meets Your Bladder
Researchers are now feeding exosomal RNA data into AI models to catch what the human eye misses. A study published in bioRxiv used a logistic regression machine learning model on urinary exosomal miRNAs (let-7c cluster). The model achieved an 80% accuracy and a 100% recall rate in distinguishing cancer patients from healthy controls, meaning it didn’t miss a single case of cancer in that trial cohort .
Part 4: Beyond Cancer (Lower Urinary Tract Dysfunction)
While bladder cancer is the headline act, exosomal RNA is also being used to investigate non-cancerous bladder diseases.
Bladder Outlet Obstruction (BOO) and Neurogenic Lower Urinary Tract Dysfunction (NLUTD) are currently diagnosed using urodynamics—a test that requires catheters in the bladder and rectum. It is uncomfortable and carries a risk of infection .
Researchers have successfully developed optimized protocols to isolate exosomes from 50ml urine samples specifically to look for miRNA signatures of fibrosis and obstruction . The goal is to replace the invasive pressure test with a simple urine RNA analysis to determine if a patient needs surgery.
Part 5: The Challenges (Why Isn’t It in Your Doctor’s Office Yet?)
If exosomes are so great, why can’t you buy this test on Amazon yet? There are three major hurdles the industry is currently solving :
- Isolation Standards: There is no universal “best way” to fish exosomes out of urine. Some labs use ultracentrifugation, others use kits or filtration. Different methods yield different results.
- The “THP” Problem: Urine contains a massive protein called Tamm-Horsfall protein (THP) that traps exosomes. If you don’t remove THP, you lose the exosomes. New protocols using detergents like CHAPS and DTT are solving this .
- Normalization: How much RNA is “normal”? Because hydration levels change how concentrated urine is, scientists are still fighting over the best way to “normalize” the data (usually using reference genes like ALDOB) .
Part 6: The Future Landscape
The field is moving toward multi-omics—combining different types of data for a perfect snapshot.
The “Liquid Biopsy” Panel:
Future tests will likely combine:
- Exosomal mRNA (for active tumor markers).
- Exosomal lncRNA (for early detection fingerprints).
- Exosomal miRNA (for recurrence risk).
Clinical Availability:
Several commercial labs are already offering “Exosome-Based Liquid Biopsy” tests for urologic cancers, though most are currently classified as Laboratory Developed Tests (LDTs) rather than over-the-counter kits. Given the meta-analysis data showing an AUC of 0.87 for multi-panel exosome tests , it is highly probable that the FDA will clear a specific urine exosome test for bladder cancer surveillance within the next 3 to 5 years.
Conclusion: The End of the Scope?
For the millions of patients living with the fear of bladder cancer recurrence, the prospect of a painless, accurate urine test is revolutionary. Exosomal RNA represents a paradigm shift: we are no longer looking for the cells themselves, but listening to the messages they send.
While we wait for these tests to hit the mainstream market, the science is clear. The future of bladder health is not a camera in the urethra—it is a genetic whisper in a plastic cup.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Exosomal RNA tests are primarily used in clinical trials and specialized research settings as of 2025. Always consult a urologist for diagnosis and treatment of bladder disease.
If you are a researcher, clinician, or patient curious about exosomal RNA, the questions above represent the cutting edge of a field that is moving from the laboratory to the clinic. The most pressing questions for the average person are likely:
- “Is there a test I can buy today?” (Mostly no, but clinical trials exist.)
- “When will this be available at my doctor’s office?” (Likely 3-7 years for widespread FDA clearance.)
- “Is it better than what I have now?” (For bladder cancer surveillance, early data says yes—especially for avoiding unnecessary cystoscopies.)
Part 1: Foundational Questions (What & Why)
- What exactly is exosomal RNA? RNA molecules packaged inside tiny extracellular vesicles called exosomes.
- How is exosomal RNA different from regular “free-floating” RNA? It is protected by a lipid membrane; free RNA degrades quickly.
- Why do cells package RNA into exosomes? For intercellular communication, waste removal, and signaling.
- What types of RNA are found inside exosomes? mRNA, miRNA, lncRNA, tRNA, rRNA, and other non-coding RNAs.
- Are exosomal RNA sequences identical to parent cell RNA? No, they are selectively packaged and often enriched for specific sequences.
- What is the biological purpose of exosomal RNA transfer? To alter gene expression in recipient cells.
- Can exosomal RNA influence recipient cells? Yes, it can be translated into protein or regulate gene expression.
- How stable is exosomal RNA? Very stable—protected from RNase enzymes for hours to days.
- Why does the lipid bilayer protect RNA? It acts as a physical barrier against degradation.
- Do all cell types produce the same exosomal RNA? No, profiles vary by cell type, health, and environment.
Part 2: Technical & Laboratory Questions (How)
- How do scientists isolate exosomes? Ultracentrifugation, size-exclusion chromatography, or precipitation kits.
- Difference between methods? Ultracentrifugation is gold standard but slow; kits are faster but less pure.
- How to remove THP from urine? Use reducing agents like DTT or CHAPS detergent.
- Optimal urine volume? 50mL is typical; some methods work with as little as 5mL.
- Can exosomal RNA come from frozen samples? Yes, but fresh or properly frozen is better.
- Methods to quantify exosomal RNA? qPCR, next-generation sequencing (NGS), microarrays, or bioanalyzer.
- Can sequencing detect 18–22 nucleotide miRNAs? Yes, small RNA sequencing is designed for this.
- Minimum concentration for detection? As low as 1–10 picograms per microliter with amplification.
- How to distinguish exosomal RNA from other vesicle RNA? By size, density, and surface markers (CD9, CD63, CD81).
- Controls to confirm RNA is inside exosomes? Treat with RNase before and after lysing exosomes.
- Can exosomal RNA be detected with biosensors? Yes, electrochemical and optical biosensors are in development.
- Role of machine learning? Identifies complex patterns and predictive signatures in large datasets.
- How to normalize for hydration? Use reference genes like ALDOB or creatinine normalization.
Part 3: Bladder Disease & Urology Questions
- Why is urine ideal for bladder exosomes? Bladder cells shed exosomes directly into stored urine.
- Do bladder cancer cells release more exosomes? Yes, typically more than normal cells.
- What miRNA signatures are associated with NMIBC? miR-21, miR-200 family, let-7c cluster.
- Can exosomal RNA distinguish low-grade from high-grade? Yes, specific signatures correlate with grade.
- Sensitivity for carcinoma in situ (CIS)? Emerging data suggests better than cytology, but not perfect.
- Can it predict recurrence? Yes, elevated KRT17 and certain miRNAs predict higher recurrence risk.
- How does profile change with progression? Loss of protective miRNAs, gain of oncogenic lncRNAs.
- Useful for upper tract tumors? Possibly, but dilution from kidney passage is a challenge.
- Differentiate cancer from benign conditions? Yes, cancer signatures are distinct from cystitis.
- What lncRNAs are validated for early cancer? CCDC148-AS1, XLOC_006419, RP5-1148A21.3.
- Accuracy vs. urine cytology? Exosomal RNA is significantly more sensitive (75% vs. ~40-50%).
- Monitor BCG or chemo response? Yes, decreasing tumor-specific RNA indicates response.
- Markers for neurogenic dysfunction? Under investigation; miRNA signatures of fibrosis and inflammation.
- Detect fibrosis/obstruction? Possible; research is ongoing to replace urodynamics.
- Signatures for recurrent UTIs? Yes, altered Lactobacillus-associated RNA profiles.
Part 4: Other Diseases & Body Fluids
- Prostate cancer detection vs. PSA? Comparable or better specificity; reduces false positives.
- Kidney disease markers? miRNAs for AKI, diabetic nephropathy, and IgA nephropathy exist.
- CSF for Alzheimer’s/Parkinson’s? Yes, exosomal α-synuclein and tau are promising.
- Saliva for head and neck cancers? Yes, highly feasible for oral squamous cell carcinoma.
- Pancreatic cancer detection? Exosomal GPC1 and specific miRNAs show early promise.
- COVID-19 severity markers? Yes, inflammatory miRNA signatures correlate with severity.
- Amniotic fluid for fetal defects? Research stage; potential for non-invasive prenatal testing.
- Endometriosis or ovarian cancer? Yes, exosomal miRNAs are under active investigation.
- Sputum for lung cancer? Feasible, but blood and pleural fluid are more common.
- Tears for eye diseases? Yes, diabetic retinopathy and dry eye signatures exist.
Part 5: Clinical & Diagnostic Questions
- Is exosomal RNA a liquid biopsy? Yes, it is a core component of liquid biopsy.
- Which tests are FDA-approved? Very few; most are CLIA-certified LDTs, not FDA cleared.
- Cost and insurance coverage? $200–$1000; generally not covered, considered investigational.
- Turnaround time for results? 5–14 days depending on lab and sequencing depth.
- Replace cystoscopy entirely? Not yet; may reduce frequency of invasive procedures.
- PPV and NPV? Varies; studies show ~80-90% for both in high-risk populations.
- How often for recurrence surveillance? Every 3-6 months if used, but not yet standard.
- Population-wide screening? Not yet; too expensive and not validated for low-risk groups.
- False positive/negative rates? Approximately 10-20% depending on the study and cutoff.
- Correlation with tumor stage/grade? Strong correlation; higher-grade tumors shed more distinct RNA.
- Detect minimal residual disease (MRD)? Yes, promising for post-surgical monitoring.
- Predict immunotherapy response? Early data suggests yes, via immune-related exosomal RNA.
Part 6: Biological & Mechanistic Questions
- How are RNAs selected for packaging? By RNA-binding proteins (hnRNPs, YB-1) recognizing specific motifs.
- What role do RNA-binding proteins play? They act as “barcodes” guiding RNA into exosomes.
- Do healthy and cancer exosomes differ? Dramatically; cancer exosomes have oncogenic and proliferative RNAs.
- Can cancer exosomal RNA transform healthy cells? In lab studies, yes—can induce proliferation and migration.
- Role in tumor microenvironment? Promotes angiogenesis, immune suppression, and metastasis.
- Transfer drug resistance? Yes, exosomes can carry resistance-conferring mRNAs and miRNAs.
- How does the immune system respond? Mixed; can activate or suppress immunity depending on context.
- Can gut microbiome exosomes reach distant organs? Theoretical; evidence is emerging but limited.
- Do diet or exercise change exosomal RNA? Yes; both alter circulating exosomal miRNA profiles.
- Does exosomal RNA get translated in recipient cells? Yes, exogenous exosomal mRNA can produce functional protein.
Part 7: Comparative Questions (vs. Other Biomarkers)
- vs. ctDNA? RNA provides functional information; ctDNA provides mutation status. Complementary.
- Stability in urine vs. cell-free DNA? Exosomal RNA is more stable due to membrane protection.
- vs. NMP22 or UroVysion? Exosomal RNA generally has higher sensitivity for low-grade tumors.
- vs. cytology for low vs. high grade? Exosomal RNA wins for low-grade; cytology still decent for high-grade.
- Information not in ctDNA? RNA splicing variants, non-coding regulation, and real-time expression.
- vs. circulating tumor cells (CTCs)? Easier to isolate, more abundant, and less fragile than CTCs.
- Cost vs. tumor tissue sequencing? Exosomal RNA is cheaper and non-invasive but less comprehensive.
Part 8: Challenges & Limitations Questions
- Biggest technical hurdles? Lack of standardization and efficient isolation methods.
- Degradation during transport? Minimal if stored cold; exosomes are stable for days at 4°C.
- Does hematuria interfere? Yes, blood-derived exosomes can contaminate results.
- Do UTIs alter profiles? Yes, bacterial infection changes host exosomal RNA signatures.
- Diurnal or hydration effects? Yes; first morning void is often preferred for consistency.
- Contamination from kidney or blood exosomes? Yes, a major challenge for bladder-specific detection.
- Inter-laboratory variability? Significant; different kits and protocols yield different results.
- Do different kits produce different results? Yes, this is a well-documented problem.
- Limit of detection for small tumors? Approximately 1–5mm tumors can be detected with optimized assays.
- Distinguish primary bladder from metastasis? Sometimes; requires tumor-specific mutation tracking.
Part 9: Future & Emerging Questions
- Home test within 5 years? Possibly; companies are developing lateral flow and smartphone-based assays.
- Guide personalized medicine? Yes, to select chemo or targeted therapy based on exosomal RNA.
- Exosomal RNA vaccines or therapeutics? Yes, engineered exosomes as drug delivery vehicles are in trials.
- Synthetic exosomes for therapy? In development for cancer and neurodegenerative diseases.
- Will multi-omics become standard? Likely yes; RNA + protein + lipid = more accurate diagnosis.
- Single urine sample for multiple cancers? Theoretical; organ-specific signatures would be needed.
- Predict preterm birth or preeclampsia? Yes, placental exosomal RNA in maternal blood shows promise.
- AI improve interpretation? Yes, deep learning is already identifying novel signatures.
- Pre-symptomatic neurodegeneration detection? Possibly 5–10 years before symptoms.
Part 10: Ethical, Regulatory & Commercial Questions
- Who owns your exosomal RNA data? Typically the lab or test manufacturer per terms of service.
- Incidental findings possible? Yes, mutations for other diseases could theoretically appear.
- Counseling for uncertainty? Patients should understand these are not yet FDA-approved diagnostics.
- Direct-to-consumer tests available? Very few; most require a physician order.
- FDA regulatory pathway? Likely Class II (moderate risk) with special controls.
- Leading companies? Exosome Diagnostics (Dx), Bio-Techne, Illumina, Evox Therapeutics.
- Patents shaping competition? Yes, fierce patent battles exist over isolation and sequencing methods.
- Will insurance replace cystoscopy with exosome tests? Possibly if large cost-effectiveness trials succeed.
Part 11: Patient & Consumer Questions
- Is exosomal RNA testing painful? No—just a blood draw or urine collection.
- How to prepare? First morning urine is ideal; no fasting required generally.
- Can I test during period or UTI? Avoid; both can alter results.
- Wait time after biopsy? At least 2–4 weeks to avoid post-procedure inflammation artifacts.
- Positive test = cancer? No; false positives exist. Requires confirmatory cystoscopy.
- Negative test = skip cystoscopy? No; false negatives exist. Follow doctor’s advice.
- Out-of-pocket cost? Approximately $300–$1500 depending on depth of analysis.
- Patient assistance programs? Rare; some companies offer sliding scale fees.
- Order yourself or need prescription? Most require a physician’s order (CLIA regulations).
- Questions for urologist? “Is this test validated for my condition? What is the false negative rate?”
Part 12: Trivia & “Out There” Questions
- Detected in sweat, tears, breast milk? Yes, exosomes are present in all these fluids.
- Plant exosomes affect humans? Possibly; plant exosomal RNAs modulate human immune responses.
- Fetal exosomes in mother’s urine? Theoretical; placental exosomes are in blood, less studied in urine.
- Exosomes in placebo effect? No evidence; highly unlikely.
- Forensic use? Theoretically yes to identify tissue origin of a stain.
- “Edit” exosomal RNA in real-time? Science fiction for now; future possibility with CRISPR-based tools.
Quick Summary Table
| Category | Most Important Takeaway |
|---|---|
| What it is | Protected RNA in tiny vesicles for cell communication |
| Best fluid for bladder | Urine (direct shedding from bladder wall) |
| Key advantage | Non-invasive, stable, functionally informative |
| Current status | Research and CLIA labs; few FDA approvals |
| Main challenge | No standardization across labs |
| Future timeline | Clinical use in 3–7 years for bladder cancer |
Disclaimer: These answers reflect the state of exosomal RNA research as of 2025. Always consult a physician for medical decisions.