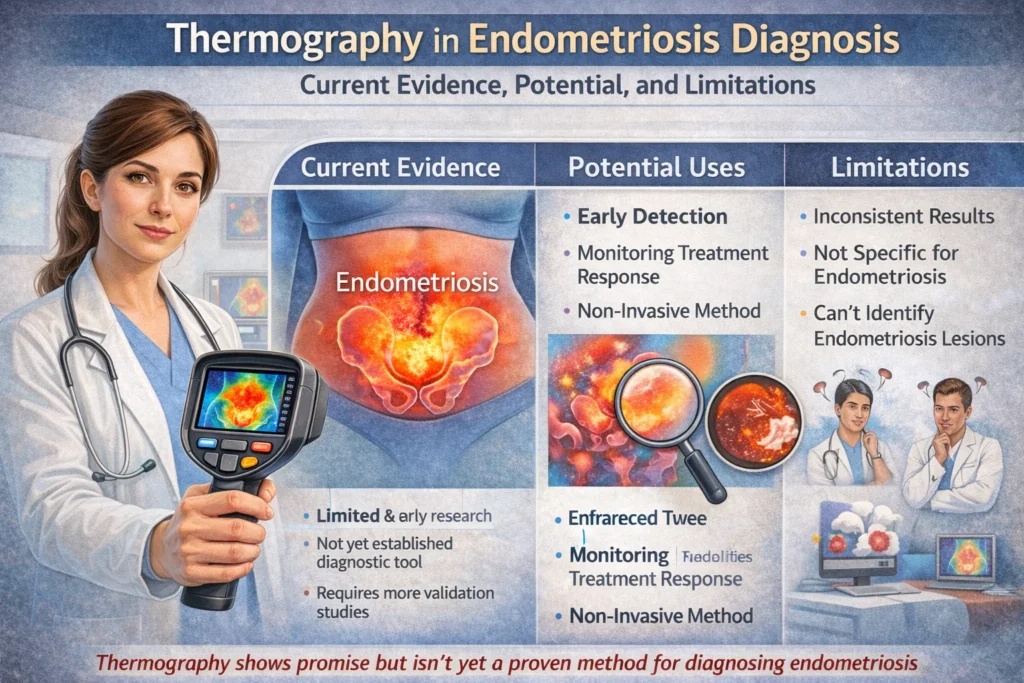
Endometriosis is a chronic condition where tissue similar to the uterine lining grows outside the uterus, often causing severe pelvic pain, inflammation, infertility, and reduced quality of life. Diagnosis is frequently delayed by 7+ years due to the need for invasive laparoscopy (the current gold standard) and the limitations of standard imaging. Thermography (infrared thermography or digital infrared thermal imaging — DITI) has been explored as a non-invasive option because endometriotic lesions trigger sterile inflammation, increased blood flow, and local heat that can appear as “hot spots” on thermal images.
How Thermography Could Relate to Endometriosis
Thermography uses sensitive infrared cameras to detect subtle skin temperature variations without contact, radiation, or discomfort. Inflamed or highly vascular endometriotic implants (e.g., in the cul-de-sac, bladder wall, bowel, or abdominal scars) may produce detectable heat patterns due to metabolic activity and vasodilation.
- Procedure: Patients acclimate in a controlled room (typically 22–24°C). Images are taken of the lower abdomen, pelvis, and back. Color-coded thermograms highlight asymmetric or elevated temperatures.
- Potential uses: Pre-screening for suspected endometriosis, mapping painful/inflamed areas, monitoring treatment response (e.g., reduction in heat after medication or surgery), or evaluating scar endometriosis (abdominal wall endometriomas post-C-section).
Case reports and small studies have shown thermographic changes in abdominal wall endometriosis, with hot spots correlating to histologically confirmed lesions.
Reported Evidence and Performance
A 2025 observational study involving over 2,000 patients (with surgical confirmation in a subset) suggested promising results when using high-sensitivity medical thermography:
- Overall accuracy: ~90% in cases without significant interfering abdominal inflammation; 70–80% when inflammation from other causes was present.
- Symptom correlation: 80–90% agreement between patient-reported pain locations and thermographic findings.
- Site-specific performance (based on ROC analysis and comparison to laparoscopy):
- Cul-de-sac lesions: Near-perfect (AUC 0.98, accuracy ~98%)
- Colon wall lesions: Excellent (AUC 0.99, accuracy ~98.5%)
- Bladder wall lesions: High (AUC 0.91, accuracy ~94%)
- Adenomyosis (intrauterine): Moderate (AUC 0.72, accuracy ~89%, lower specificity due to fibroids mimicking patterns)
- Follow-up scans showed correlation between symptom improvement and reduced thermal signals (statistically significant).
The authors proposed thermography as a pre-screening and monitoring tool rather than a definitive diagnostic, potentially reducing reliance on repeated surgeries. They noted benefits when combined with ultrasound or enhanced by AI for pattern recognition.
Other limited evidence includes case reports on scar endometriosis where thermography helped visualize localized heat not fully explained by physical exam or ultrasound alone.
Comparison to Standard Imaging for Endometriosis
Standard non-invasive tools focus on anatomical visualization:
- Transvaginal Ultrasound (TVUS): First-line, widely available, real-time. Excellent for ovarian endometriomas and some deep infiltrating endometriosis (DIE) when performed by experts using specialized protocols. Sensitivity and specificity vary by location (often 70–90%+ for rectosigmoid). Operator-dependent.
- MRI: Often superior for deep infiltrating endometriosis, global pelvic assessment, and extrapelvic disease. High sensitivity/specificity (typically 80–95% for many sites) with dedicated protocols. Better for complex cases but more expensive and less accessible.
- Thermography: Provides physiological (heat/inflammation) rather than structural data. It cannot visualize lesion size, depth, or exact anatomy like ultrasound or MRI.
Key differences:
- Thermography is completely non-contact and painless — ideal for patients with severe pain or who cannot tolerate internal probes.
- It may detect early inflammatory changes but lacks specificity (heat can result from infections, cysts, fibroids, muscle strain, or other conditions).
- Ultrasound and MRI remain far more established and accurate for confirming structural endometriosis and guiding surgery.
Major Limitations and Expert Consensus
Despite some encouraging preliminary data, thermography is not validated or recommended as a diagnostic tool for endometriosis by major medical bodies:
- Low specificity and confounding factors: Heat patterns can be altered by bowel inflammation, infections, recent activity, room temperature, or other pelvic conditions, leading to false positives or negatives.
- Limited high-quality evidence: Most data come from small studies, case reports, or single-center observations. Large, prospective, multicenter trials with standardized protocols are lacking. Many reviews and clinical guidelines do not include thermography as a reliable method.
- Not a replacement for gold standard: Laparoscopy with histological confirmation is still required for definitive diagnosis in many cases. Thermography cannot reliably differentiate endometriosis from other causes of pelvic pain or inflammation.
- Availability and regulation: Often offered in wellness or integrative clinics rather than mainstream medical settings. Results depend heavily on equipment quality, technician expertise, and interpretation.
Experts emphasize that thermography should not be used in isolation for diagnosis, as it may delay proper evaluation with proven imaging or surgical assessment.
Practical Takeaways
- Potential role: Thermography may serve as a safe, low-cost adjunct for monitoring inflammation or guiding further targeted testing (e.g., prompting expert ultrasound/MRI). It could be particularly useful in scar endometriosis or for tracking treatment effects non-invasively.
- When to consider standard care: If you have symptoms suggestive of endometriosis (cyclic pelvic pain, painful periods, pain with intercourse, infertility), consult a gynecologist or endometriosis specialist. They will typically start with clinical history, exam, and dedicated transvaginal ultrasound, progressing to MRI or laparoscopy as needed.
- Future outlook: Integration with AI, combination with ultrasound, or improved protocols might strengthen its utility, but more rigorous research is required.
Disclaimer: This information is for educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment. Thermography is not currently a validated or recommended primary method for diagnosing endometriosis. Always consult a qualified healthcare provider (such as a gynecologist or pelvic pain specialist) for personalized evaluation. Do not rely on thermography or any single test in isolation for health decisions. Early and accurate diagnosis through established methods can significantly improve outcomes.
If you’re experiencing pelvic symptoms, seek medical attention promptly rather than depending on experimental or adjunctive techniques. Would you like this integrated with previous articles on pelvic floor health, thermography basics, or comparisons to ultrasound?