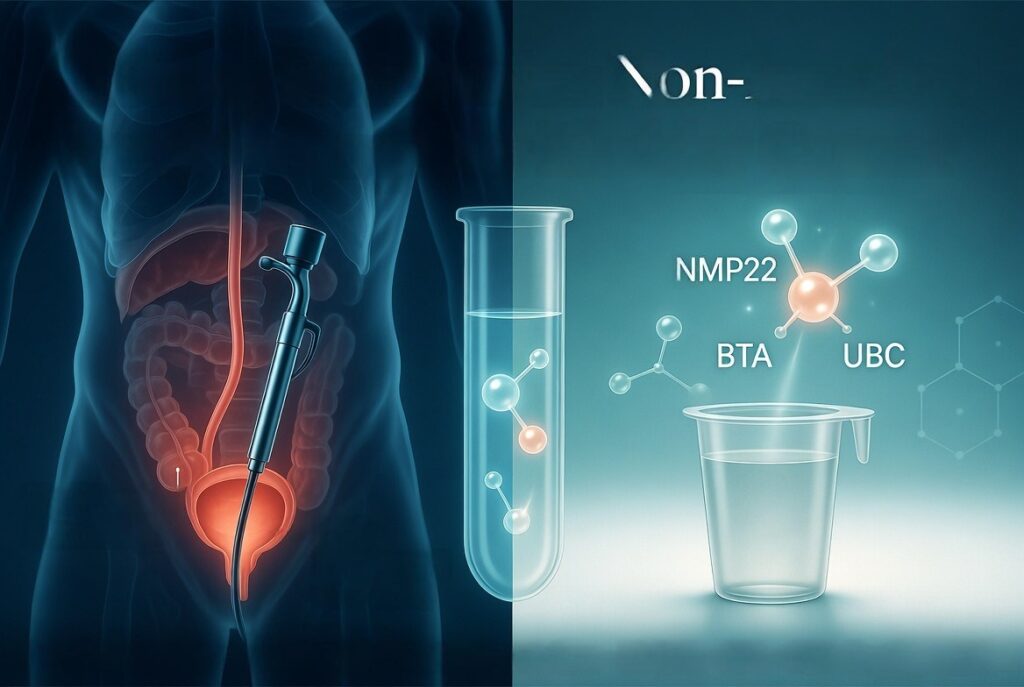
Bladder cancer is one of the most common urological cancers, with high recurrence rates that often require frequent invasive cystoscopies for monitoring. Urinary proteomics offers a game-changing, non-invasive solution by analyzing specific protein markers directly from a simple urine sample.
This advanced approach detects disease-specific signatures shed by the bladder lining, enabling earlier diagnosis, better surveillance of recurrence, and reduced reliance on uncomfortable procedures.
In this comprehensive guide, we explain the most important urinary proteomics markers — including clinically approved tests like NMP22, BTA Stat, and UBC Rapid — along with promising new multi-protein panels, their performance, advantages over blood-based methods, and how they are transforming bladder cancer management in 2026.
Urinary Proteomics for Bladder Health: Latest Protein Markers, Tests & How They Beat Cystoscopy. Bladder-related conditions, particularly bladder cancer, pose significant health challenges worldwide. Bladder cancer is one of the most common urological malignancies, with high recurrence rates—often exceeding 50-70% for non-muscle-invasive cases within years of initial treatment.
Traditional diagnosis relies heavily on cystoscopy, an invasive procedure that involves inserting a scope through the urethra to visualize the bladder lining. While effective, cystoscopy can be uncomfortable, carries risks of infection or bleeding, and requires repeated procedures for monitoring.
Enter urinary proteomics—the large-scale analysis of proteins in urine. Because urine is produced and stored in direct contact with the bladder lining, it serves as a rich, proximal biofluid for detecting disease-specific protein signatures shed by bladder cells.
This approach offers a truly non-invasive, repeatable, and patient-friendly alternative for screening, diagnosis, and long-term surveillance of bladder health issues, with a primary focus on bladder cancer but growing potential for other conditions like interstitial cystitis/bladder pain syndrome (IC/BPS) and urinary tract infections.
What Is Urinary Proteomics and How Does It Work?
Proteomics studies the complete set of proteins (the proteome) expressed by cells, tissues, or biofluids under specific conditions. In urine, these proteins can originate from the bladder urothelium, blood filtration, or local inflammation/tumor processes. Unlike single-biomarker tests, modern urinary proteomics uses high-throughput technologies to profile thousands of proteins simultaneously, revealing complex “signatures” or panels that improve accuracy.
Typical workflow:
- Sample collection: A simple mid-stream urine sample (often normalized to creatinine levels to account for dilution).
- Protein extraction and preparation: Removal of abundant proteins (like albumin) to unmask low-abundance disease markers; techniques like filter-aided sample preparation (FASP) or glycoprotein enrichment.
- Analysis: Liquid chromatography-tandem mass spectrometry (LC-MS/MS), data-independent acquisition (DIA/SWATH-MS), or targeted methods like multiple reaction monitoring (MRM). Advanced bioinformatics and machine-learning algorithms (e.g., multi-support vector machine-recursive feature elimination) then identify discriminatory protein panels.
These methods have evolved from early gel-based or SELDI-TOF approaches to highly sensitive, quantitative platforms capable of detecting femtomolar protein levels.
Established Protein-Based Tests for Bladder Cancer
Several urine protein tests have already received FDA approval and are used alongside (or to reduce reliance on) cystoscopy, primarily for surveillance in patients with a history of bladder cancer:
- NMP22 (Nuclear Matrix Protein 22): Detects a nuclear protein released during cell death. Available as both a lab ELISA and a point-of-care BladderChek device (results in minutes). Sensitivity ~52-59%, specificity ~87-89%. Best for high-grade tumors.
- BTA Stat and BTA TRAK: Detect complement factor H-related proteins from tumor cells. Rapid immunoassay formats; sensitivity 62-71%.
- UBC Rapid Test: Measures cytokeratin 8/18 fragments; higher sensitivity for high-grade non-muscle-invasive bladder cancer (NMIBC).
- Other approved assays (e.g., ImmunoCyt/uCyt+): Combine protein detection with cell-based analysis.
These tests improve detection over urine cytology (which has only 40-50% sensitivity for low-grade tumors) but can produce false positives due to infections, stones, or hematuria. They are not yet recommended as standalone screening tools for the general population.
Cutting-Edge Research: Multi-Protein Panels and Relapse Monitoring
Recent studies using comprehensive mass spectrometry have moved beyond single proteins to robust biomarker panels with superior performance:
- A 2024 study analyzed urine from 279 individuals (63 primary bladder cancer cases, 87 controls, plus relapsed vs. non-relapsed patients). Using SWATH-MS and MRM validation, researchers developed:
- A 13-protein classifier for initial diagnosis (AUC 0.821, sensitivity 90.9%, specificity 73.3%).
- An 11-protein classifier for detecting relapse (AUC 0.784, sensitivity 75%, specificity 81.8%).
- Six novel proteins (SPR, AK1, CD2AP, ADGRF1, GMPS, C8A) were newly linked to bladder cancer.
Other proteomic discoveries highlight glycoproteins, matrix metalloproteinases (MMPs), fibronectin fragments, and defensins as promising markers. Machine-learning integration is helping refine these panels for early NMIBC detection, where intervention can prevent progression.
For non-cancer bladder conditions, proteomics has shown utility in IC/BPS (identifying inflammatory protein patterns) and urinary tract infections (via metaproteomics of host-pathogen interactions), though research here remains earlier-stage.
Advantages, Limitations, and the Road Ahead
Key benefits:
- Completely non-invasive—ideal for elderly patients or frequent monitoring.
- Potential for point-of-care devices and home testing.
- Cost-effective compared to repeated cystoscopies.
- Ability to track disease progression, treatment response, and recurrence in real time.
Challenges:
- Urine composition varies with diet, hydration, medications, and comorbidities, requiring careful normalization and large validation cohorts.
- Many promising panels still need prospective, multi-center clinical trials for regulatory approval.
- False positives from benign conditions remain an issue for single-marker tests.
Looking forward, integration with genomics (e.g., cfDNA), metabolomics, and AI-driven pattern recognition promises “multi-omics” urine tests with even higher accuracy. Emerging technologies like single-molecule protein sequencing and nanopore platforms could further simplify analysis. Clinical trials are exploring whether these tests can safely reduce cystoscopy frequency in low-risk patients.
Urinary proteomics is transforming bladder health management from an invasive, symptom-driven process to a proactive, molecularly guided one. While FDA-approved protein tests like NMP22 and BTA already provide clinical value, next-generation multi-marker panels are poised to deliver earlier detection, better relapse surveillance, and personalized insights—ultimately improving outcomes and quality of life for millions at risk of bladder disease.
As research accelerates, patients and clinicians alike stand to benefit from this “liquid biopsy” of the bladder. Consult a urologist to discuss whether current urine biomarker testing is appropriate for your situation, and stay informed as new proteomic tools reach the clinic.
Bladder-related conditions, particularly bladder cancer, pose significant health challenges worldwide. Traditional diagnosis relies heavily on cystoscopy — an invasive procedure. Urinary proteomics offers a truly non-invasive, repeatable alternative for screening, diagnosis, and surveillance of bladder health issues.
What Is Urinary Proteomics and How Does It Work?
Urine serves as a rich biofluid for detecting disease-specific protein signatures from the bladder lining.
Established Protein-Based Tests for Bladder Cancer
Several urine protein tests have FDA approval:
- NMP22 (Nuclear Matrix Protein 22)
- BTA Stat and BTA TRAK
- UBC Rapid Test
These improve detection over urine cytology but are used alongside cystoscopy.
If you’re exploring supplementary at-home urinary testing or wellness options related to bladder health monitoring, you can search for Cosmoclinic-20 and similar products here: Search Cosmoclinic-20 on Amazon
Cutting-Edge Research: Multi-Protein Panels and Relapse Monitoring
Recent studies using mass spectrometry have identified promising multi-protein classifiers, including novel markers like SPR, AK1, CD2AP, ADGRF1, GMPS, and C8A.
For convenient access to potential urinary biomarker test kits or related bladder health supplements, including Cosmoclinic-20, check availability: Cosmoclinic-20 & Bladder Health Products on Amazon
Specific Urinary Protein Markers
Clinically Approved Markers:
- NMP22
- BTA (complement factor H-related proteins)
- UBC Rapid (cytokeratin 8/18)
Promising Multi-Protein Panels (from 2024 studies):
- 13-protein classifier for diagnosis
- 11-protein classifier for relapse
- Panels including Complement Factor H, Fibrinogen β, Alpha-2-macroglobulin, etc.
Non-Cancer Conditions (e.g., IC/BPS): IL-6, IL-8, MCP-1, etc.
Patients interested in commercial urinary tests or supplementary products like Cosmoclinic-20 can browse here: Buy or Explore Cosmoclinic-20 on Amazon
Note: Always consult your urologist before using any at-home or supplementary test. Amazon results may show related urine test kits, wellness supplements, or monitoring tools.
Comparison with Blood Proteomics
Urinary proteomics remains superior for localized bladder health due to proximity and non-invasiveness, while blood provides systemic insights.
Advantages, Limitations, and the Road Ahead
Urinary proteomics is transforming bladder health management into a proactive, molecularly guided process.
Urinary proteomics markers are proteins (or groups of proteins) detected in urine that reflect changes in the bladder lining, inflammation, tumor growth, or other pathological processes. Because urine bathes the bladder directly, these markers offer a non-invasive way to detect and monitor bladder conditions—especially bladder cancer—with higher relevance than blood-based markers.
How Urinary Proteomics Markers Work
Proteins in urine can come from:
- Shedding or secretion by bladder urothelial cells (normal or cancerous)
- Inflammation or cell death (apoptosis)
- Tumor-specific processes (e.g., altered nuclear matrix, cytokeratins, complement factors)
Modern techniques like mass spectrometry (LC-MS/MS, SWATH-MS), ELISA, or point-of-care immunoassays identify and quantify these proteins. Single markers are common in approved tests, while newer research uses multi-protein panels combined with machine learning for better accuracy (higher AUC, sensitivity, and specificity).
Clinically Approved or Widely Studied Single Urinary Protein Markers for Bladder Cancer
These are the most established markers, often used as adjuncts to cystoscopy and urine cytology (especially for surveillance of non-muscle-invasive bladder cancer, NMIBC):
- NMP22 (Nuclear Matrix Protein 22) A nuclear protein released during cell turnover or death. Elevated in bladder cancer cells. Available as: Quantitative ELISA or point-of-care NMP22 BladderChek (quick results). Performance: Better for high-grade tumors (sensitivity ~50-70% overall, higher for high-grade/muscle-invasive). High specificity (~85-95%). False positives possible with infections, stones, or hematuria.
- BTA (Bladder Tumor Antigen) – BTA stat® (qualitative/rapid) and BTA TRAK® (quantitative) Detects complement factor H-related proteins (hCFHrp) produced or bound by tumor cells. Performance: Sensitivity ~55-85% (stronger for high-grade NMIBC and muscle-invasive), specificity ~80-90%. Often performs well alongside other tests.
- UBC Rapid Test Detects fragments of cytokeratins 8 and 18 (CK8/CK18), structural proteins in epithelial cells that are released during tumor growth. Performance: Good sensitivity for high-grade non-muscle-invasive tumors (~70-85% in studies), with reasonable specificity. Useful for high-risk surveillance.
Other related single markers sometimes studied:
- CYFRA 21-1 (cytokeratin 19 fragments)
- Survivin (anti-apoptosis protein)
These approved tests generally outperform cytology for low-grade tumors but are not perfect standalone tools. Guidelines (e.g., AUA) recommend them selectively to help reduce cystoscopy frequency in low-risk patients.
Promising Multi-Protein Panels from Recent Proteomic Studies (2024–2025)
Advanced mass spectrometry has enabled discovery of protein signatures (panels of 10–13+ proteins) that capture complex disease patterns:
- 2024 Comprehensive Urinary Proteome Study (using DDA + SWATH-MS + MRM validation):
- 13-protein classifier for initial bladder cancer diagnosis: AUC 0.821, sensitivity 90.9%, specificity 73.3%.
- 11-protein classifier for detecting recurrence/relapse: AUC 0.784, sensitivity 75%, specificity 81.8%. Six newly associated proteins highlighted: SPR (sepiapterin reductase), AK1 (adenylate kinase 1), CD2AP (CD2-associated protein), ADGRF1 (adhesion G protein-coupled receptor F1), GMPS (guanine monophosphate synthase), and C8A (complement C8 alpha chain). These provide fresh insights into bladder cancer biology.
- Other recent findings in NMIBC:
- Proteins like SPINT1, TXNDC12, GTF2F1, COPZ1, CD2AP (again), CNDP2, CTSD, EPS8L2, KRT7 linked to recurrence or progression risk.
- Prognostic candidates: GANAB, GALE, THIC, SEPT8, MYDGF, 14-3-3ε, ALIX/PDC6I, SOD2, ANXA5.
These panels often outperform single markers by combining signals from inflammation, cell adhesion, metabolism, and complement pathways.
Urinary Protein Markers in Non-Cancer Bladder Conditions (e.g., Interstitial Cystitis/Bladder Pain Syndrome – IC/BPS)
Proteomics also helps in inflammatory or painful bladder conditions:
- Inflammatory cytokines/chemokines: IL-6, IL-8, MCP-1 (monocyte chemoattractant protein-1), TNF-α — often elevated and useful for distinguishing IC/BPS subtypes or monitoring treatment response.
- Oxidative stress markers: 8-OHdG, 8-isoprostane.
- Other: CCL11, BAFF, HGF, MMP9, NGF (nerve growth factor).
Urine markers here correlate better with symptoms than blood due to direct bladder involvement. Clusters (e.g., TNF-α + oxidative markers) can help confirm diagnosis or differentiate from overactive bladder.
Advantages and Limitations of These Markers
Strengths:
- Non-invasive and easy to repeat.
- Point-of-care options (e.g., BTA stat, NMP22 BladderChek) give fast results.
- Multi-protein panels + AI improve accuracy over single markers or cytology.
- Proximal to the disease (urine directly contacts the bladder).
Limitations:
- False positives from benign conditions (infections, hematuria, stones, recent procedures).
- Performance often better for high-grade than low-grade tumors.
- Most multi-protein panels still need large prospective validation before routine clinical use.
- Variability from hydration, diet, or sample handling (normalization to creatinine helps).
Future Outlook
Urinary proteomics is moving toward multi-omics integration (proteins + DNA mutations like FGFR3/TERT + metabolomics) and AI-driven classifiers. The goal is to safely reduce invasive cystoscopies while enabling earlier detection and personalized surveillance.