Introduction
Cancer-associated thrombosis (CAT) remains a leading cause of non-cancer mortality in women with gynaecological malignancies. Traditional clinical risk scores provide a useful baseline, but their predictive accuracy is often modest—particularly in ovarian and advanced-stage disease. The current frontier is integrated risk modeling, where circulating biomarkers are combined with clinical variables to deliver more precise, individualized VTE risk stratification.
Why Clinical Scores Alone Are Not Enough
Widely used tools like the Khorana Score rely on static variables (BMI, blood counts, cancer site). While practical, they have limitations:
- Underestimation of risk in ovarian cancer
- Poor sensitivity for dynamic changes during treatment
- Lack of mechanistic insight into hypercoagulability
This has driven the shift toward biomarker-augmented models.
Key Biomarkers in Thrombosis Prediction
1. Coagulation Activation Markers
- D-dimer: Reflects fibrin degradation; consistently the strongest predictor
- Prothrombin fragment 1+2: Indicates thrombin generation
👉 Elevated levels correlate with active clot formation and breakdown
2. Platelet Activation Markers
- Soluble P-selectin: Marker of platelet-endothelial interaction
- Platelet-derived microparticles
These reflect cellular drivers of thrombosis, not captured by routine labs.
3. Tumor-Driven Procoagulants
- Tissue factor (TF) expression
- TF-bearing extracellular vesicles
Tumors—especially ovarian—directly activate coagulation cascades, amplifying risk.
4. Inflammatory Biomarkers
C-reactive protein (CRP)
- Interleukin-6 (IL-6)
Inflammation enhances thrombosis via endothelial activation and cytokine signaling.
The Biological Framework
The synergy between biomarkers and clinical factors aligns with Virchow’s triad:
- Hypercoagulability → D-dimer, tissue factor
- Endothelial injury → inflammatory markers
- Stasis → clinical factors (immobility, tumor bulk)
👉 Combining both domains captures the full thrombotic phenotype
Integrated Prediction Models
1. Vienna CATS Model
The Vienna Cancer and Thrombosis Study model integrates:
- D-dimer
- Soluble P-selectin
- Cancer type
Strength: Demonstrates significantly improved predictive accuracy over clinical scores alone.
2. COMPASS-CAT Model
The COMPASS-CAT score combines:
- Clinical variables (comorbidities, treatment)
- Patient history
- Limited biomarker integration
Use case: Practical for outpatient oncology settings.
3. Emerging Hybrid Models
Recent studies are developing multi-parametric models combining:
- Clinical risk scores
- Biomarker panels
- Imaging findings
- Genetic predisposition
These models are moving toward real-time risk prediction.
Evidence Supporting Combined Approaches
Improved Predictive Performance
- Studies show AUC improvement from ~0.60 (clinical alone) to 0.75–0.85 with biomarker integration
- Better identification of high-risk subgroups needing prophylaxis
Dynamic Risk Assessment
Biomarkers allow:
- Monitoring during chemotherapy cycles
- Detection of early hypercoagulable states before clinical events
Practical Clinical Application
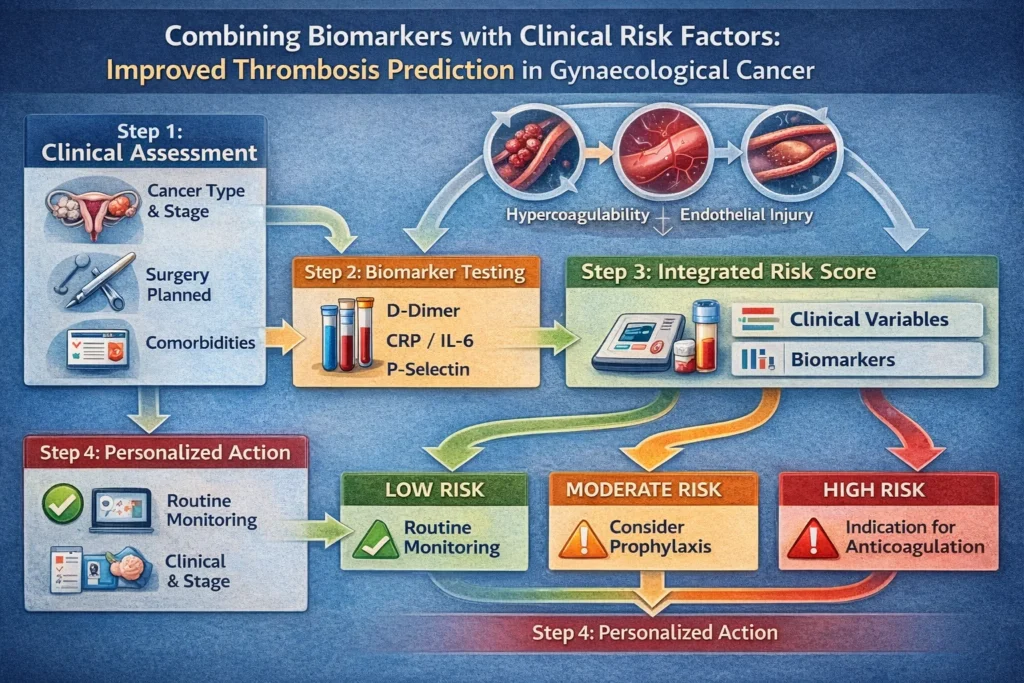
Stepwise Risk Stratification
Step 1: Baseline Clinical Assessment
- Cancer type and stage
- Surgery planned
- Comorbidities
Step 2: Biomarker Testing
- D-dimer
- CRP / IL-6
- P-selectin (where available)
Step 3: Integrated Risk Categorization
| Risk Level | Clinical + Biomarker Profile | Action |
|---|---|---|
| Low | Low clinical score + normal biomarkers | Routine monitoring |
| Intermediate | Moderate clinical risk + mild biomarker elevation | Consider prophylaxis |
| High | High clinical risk + elevated biomarkers | Strong indication for anticoagulation |
Advantages of Combined Models
- Higher sensitivity and specificity
- Early detection of subclinical thrombosis risk
- Personalized prophylaxis decisions
- Reduction in unnecessary anticoagulation (bleeding risk)
Challenges and Limitations
1. Standardization Issues
- Biomarker cutoffs vary across studies
- Limited availability of specialized assays (e.g., P-selectin)
2. Cost and Accessibility
- Advanced biomarker panels may not be widely accessible
- Resource constraints in low- and middle-income settings
3. Integration into Practice
- Lack of universally accepted guidelines
- Need for clinician education and workflow adaptation
Future Directions
1. Artificial Intelligence Integration
AI models can:
- Continuously update risk scores
- Integrate EHR + lab + imaging data
- Predict events before biomarker thresholds are crossed
2. Multi-Omics Approaches
Combining:
- Genomics
- Proteomics
- Metabolomics
This may identify novel thrombosis signatures unique to tumour biology.
3. Point-of-Care Testing
Portable biomarker devices could enable:
- Real-time monitoring
- Rapid outpatient decision-making
Key Takeaways
- Thrombosis prediction in gynaecological cancer is evolving toward integrated models
- Biomarkers such as D-dimer and P-selectin significantly enhance predictive accuracy
- Combining biological signals with clinical risk factors enables precision thromboprophylaxis
- Future models will likely be dynamic, AI-driven, and personalized
Conclusion
The integration of biomarkers with clinical risk factors represents a paradigm shift in predicting venous thrombosis in gynaecological cancer. Rather than relying on static scoring systems, clinicians can now move toward multidimensional, patient-specific risk assessment. This approach not only improves prediction but also refines prevention strategies—ultimately reducing both thrombotic complications and treatment-related harm.