The liver is the body’s silent workhorse. Weighing about three pounds, this reddish-brown organ sits in the upper right abdomen, performing over 500 vital functions—filtering toxins, producing bile for digestion, storing energy, synthesizing proteins, and regulating blood clotting. Remarkably, the liver is also the only solid organ that can regenerate itself. Even after up to 70-80% of its tissue is removed or damaged, it can grow back to its original size within weeks.
But this regenerative capacity has a limit. When injury outpaces repair—whether from chronic alcohol use, viral hepatitis, fatty liver disease, or genetic disorders—scar tissue accumulates. Eventually, the liver reaches a point of no return: end-stage liver disease (ESLD) . At this stage, the liver can no longer sustain life, and a liver transplant becomes the only remaining option.
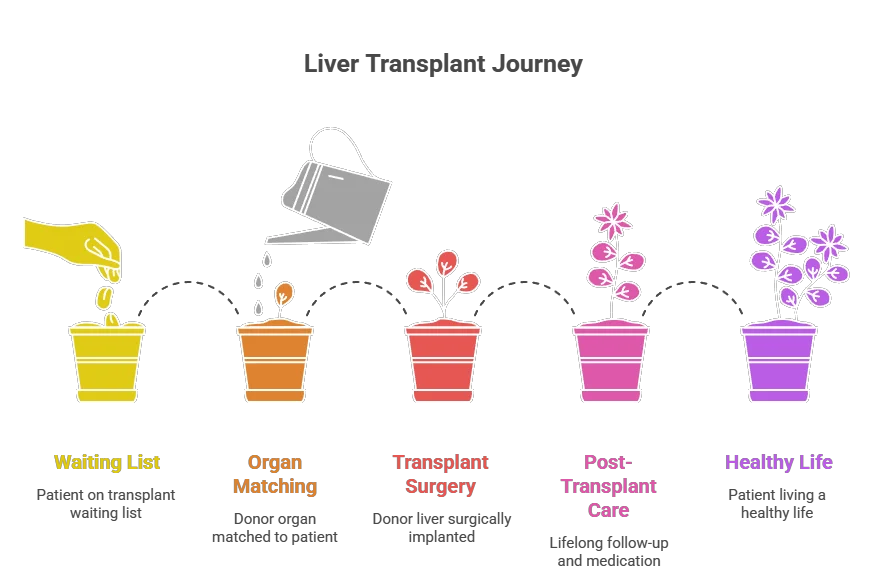
This article explains the spectrum of liver disease, the specific conditions that lead to irreversible failure, and the medical criteria that determine when transplantation is not just an option, but a necessity.
Part 1: The Spectrum of Liver Disease – From Inflammation to Cirrhosis
Liver disease is not a single condition but a continuum of injury and repair. Understanding this progression helps clarify why some patients recover with medical management while others require transplantation.
Stage 1: Hepatitis (Inflammation)
The earliest stage of liver disease is hepatitis—inflammation of the liver. Causes include:
- Viral hepatitis (Hepatitis B, C, D, E)
- Alcohol-related liver disease (ALD)
- Metabolic dysfunction-associated steatotic liver disease (MASLD) , formerly known as non-alcoholic fatty liver disease (NAFLD)
- Autoimmune hepatitis
- Drug-induced liver injury (e.g., acetaminophen overdose, certain antibiotics, herbal supplements)
At this stage, the liver is swollen but still functional. Patients may be asymptomatic or experience fatigue, nausea, or right upper quadrant discomfort. With treatment—antiviral drugs for hepatitis B/C, alcohol cessation, weight loss, or immunosuppression for autoimmune disease—inflammation often resolves without permanent damage.
Stage 2: Fibrosis (Scarring)
When inflammation persists, the liver attempts to heal by depositing collagen and other extracellular matrix proteins. This process is fibrosis—the formation of scar tissue. Unlike healthy liver tissue, scar tissue does not perform any liver functions. It also distorts the liver’s normal architecture, impeding blood flow.
Fibrosis is graded on a scale from F0 (no fibrosis) to F4 (cirrhosis). Early fibrosis (F1-F2) is potentially reversible if the underlying cause is removed. Advanced fibrosis (F3, also called bridging fibrosis) is less reversible but may still stabilize with treatment.
Stage 3: Cirrhosis (End-Stage Scarring)
Cirrhosis is the end result of longstanding, progressive fibrosis. At this stage, the liver is permanently scarred, nodular, and shrunken. The regenerative capacity is exhausted. Cirrhosis is irreversible—no medication, diet, or lifestyle change can turn scar tissue back into healthy liver cells.
Once a patient develops cirrhosis, they are at risk for two categories of life-threatening complications:
- Decompensation events (see below)
- Hepatocellular carcinoma (HCC) , the most common type of primary liver cancer
Stage 4: Decompensated Cirrhosis
Cirrhosis is classified as compensated (the liver still performs its functions despite scarring) or decompensated (liver function has failed). Decompensation is defined by the development of one or more of the following complications:
| Complication | Description | Why It Happens |
|---|---|---|
| Ascites | Accumulation of fluid in the abdominal cavity | Portal hypertension (increased blood pressure in the portal vein) forces fluid out of blood vessels into the abdomen |
| Variceal Hemorrhage | Bleeding from enlarged veins (varices) in the esophagus or stomach | Portal hypertension diverts blood to smaller, fragile veins that rupture easily |
| Hepatic Encephalopathy | Confusion, drowsiness, coma due to toxin buildup | The failing liver cannot remove ammonia and other neurotoxins from the blood |
| Jaundice | Yellowing of the skin and eyes | The liver cannot process bilirubin, a breakdown product of red blood cells |
| Coagulopathy | Bleeding tendency | The liver cannot produce enough clotting factors |
| Hepatorenal Syndrome (HRS) | Kidney failure due to liver failure | Severe portal hypertension and systemic inflammation reduce blood flow to the kidneys |
| Spontaneous Bacterial Peritonitis (SBP) | Infection of ascitic fluid | Bacteria from the gut translocate into the ascites fluid |
Once a patient develops any of these decompensation events, their median survival drops from over 12 years (compensated cirrhosis) to approximately 1-2 years. At this point, liver transplant evaluation becomes urgent.
Part 2: Specific Liver Diseases That Lead to Transplant
Not all liver diseases progress to cirrhosis and decompensation. However, the following conditions are the leading indications for liver transplantation worldwide.
1. Hepatitis C Cirrhosis (Declining but Still Significant)
Before the advent of direct-acting antivirals (DAAs) in 2014, hepatitis C was the most common indication for liver transplant in the Western world. DAAs now cure over 95% of hepatitis C cases with 8-12 weeks of oral therapy.
When does hepatitis C still require transplant?
- Patients diagnosed with advanced cirrhosis before DAA treatment became available
- Patients who develop hepatocellular carcinoma (HCC) on a background of hepatitis C cirrhosis
- Patients with decompensated cirrhosis who are cured with DAAs but have irreversible liver damage
Important nuance: Patients with decompensated hepatitis C cirrhosis can sometimes undergo DAA treatment before transplant, which may improve liver function enough to delay or avoid transplant. In other cases, they undergo transplant first, then DAA treatment after.
2. Alcohol-Related Liver Disease (ALD)
Alcohol-related liver disease is now the leading indication for liver transplantation in many countries, including the United States and Europe. It encompasses:
- Alcoholic hepatitis: Severe, acute inflammation of the liver in heavy drinkers
- Alcoholic cirrhosis: Progressive scarring due to years of heavy alcohol use
Controversy and criteria: Liver transplantation for ALD requires a strict 6-month abstinence period in most centers, along with demonstrated engagement in alcohol rehabilitation programs. However, selected patients with severe alcoholic hepatitis who fail medical therapy may be considered for “early transplant” before completing 6 months of abstinence—a practice that remains debated but is increasingly accepted.
Outcomes: Post-transplant survival for ALD is excellent, comparable to or better than other indications (approximately 85-90% 1-year survival). However, relapse to harmful drinking occurs in 10-20% of patients, which can damage the transplanted liver.
3. Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD)
Formerly called non-alcoholic fatty liver disease (NAFLD), MASLD is the hepatic manifestation of the metabolic syndrome (obesity, diabetes, hypertension, dyslipidemia). It affects approximately 25-30% of the global population.
Progression:
- Simple steatosis (MASLD without inflammation): Generally benign
- Metabolic dysfunction-associated steatohepatitis (MASH): Inflammation + fat + fibrosis
- MASH cirrhosis: Advanced scarring
- Hepatocellular carcinoma: Can occur even without cirrhosis in MASLD
Why MASLD is rising: The obesity epidemic has made MASLD the fastest-growing indication for liver transplantation, particularly in younger patients (age 40-60). Many MASLD patients also have cardiovascular disease, chronic kidney disease, and diabetes, making them higher-risk transplant candidates.
4. Acute Liver Failure (Fulminant Hepatic Failure)
Unlike chronic liver disease, acute liver failure (ALF) develops over days to weeks in a patient with no prior liver disease. It is a medical emergency with high mortality without urgent transplant.
Common causes of ALF:
- Acetaminophen overdose (most common cause in the US and UK)
- Viral hepatitis (especially Hepatitis A, B, E)
- Drug-induced liver injury (idiosyncratic reactions to antibiotics, anticonvulsants, herbal supplements)
- Autoimmune hepatitis
- Wilson’s disease (copper accumulation)
- Acute fatty liver of pregnancy
- Budd-Chiari syndrome (hepatic vein thrombosis)
Transplant criteria for ALF: Patients with ALF are listed for transplant at the highest priority (Status 1 in the US) if they meet specific criteria indicating irreversible injury, including:
- Hepatic encephalopathy (grade 3 or 4)
- INR > 6.5 (severe coagulopathy)
- Falling liver volume on imaging
- Lactate > 4 mmol/L in anhepatic phase
- Multiorgan failure
Without transplant, mortality in severe ALF exceeds 80%. With emergency transplant, 1-year survival is approximately 80-90%.
5. Primary Liver Cancers
Hepatocellular carcinoma (HCC) is the most common primary liver cancer. Not all HCC patients require transplant; early-stage HCC can be treated with:
- Surgical resection (if tumor is localized and liver function is good)
- Ablation (radiofrequency or microwave ablation)
- Transarterial chemoembolization (TACE)
- Radiation (SBRT, proton beam)
When is transplant indicated for HCC? The Milan Criteria (adopted internationally) define which HCC patients have acceptable outcomes with transplant:
- Single tumor ≤ 5 cm, or
- 2-3 tumors, each ≤ 3 cm
- No extrahepatic spread
- No major vascular invasion (portal vein, hepatic vein)
Patients within Milan Criteria have a 5-year survival after transplant of approximately 70-80% , comparable to patients transplanted for non-cancer indications. Patients outside Milan Criteria may be considered for “downstaging” (treating tumors to reduce them to within Milan Criteria) before transplant.
Cholangiocarcinoma (bile duct cancer) is a less common indication. Highly selected patients with hilar cholangiocarcinoma (Klatskin tumors) may be candidates for transplant after neoadjuvant chemoradiation, but this remains controversial and is performed at only a few specialized centers.
6. Primary Sclerosing Cholangitis (PSC) and Primary Biliary Cholangitis (PBC)
These are autoimmune cholestatic liver diseases—disorders in which the immune system attacks the bile ducts, leading to progressive bile accumulation (cholestasis), fibrosis, and cirrhosis.
Primary Sclerosing Cholangitis (PSC):
- Strongly associated with inflammatory bowel disease (IBD) , especially ulcerative colitis
- High risk of cholangiocarcinoma (bile duct cancer) and colorectal cancer
- No effective medical therapy; transplant is the only definitive treatment for end-stage PSC
Primary Biliary Cholangitis (PBC):
- Formerly called primary biliary cirrhosis
- Affects middle-aged women predominantly
- Ursodeoxycholic acid (UDCA) slows progression in many patients
- Transplant indicated for those who fail UDCA and develop decompensated cirrhosis
7. Inherited Metabolic Liver Diseases
Several genetic disorders cause progressive liver damage or life-threatening metabolic derangements. Transplant not only replaces the diseased liver but also corrects the underlying metabolic defect because the new liver produces the missing enzyme.
| Disease | Deficient Enzyme/Protein | Transplant Indication |
|---|---|---|
| Wilson’s disease | ATP7B (copper transporter) | Acute liver failure or decompensated cirrhosis unresponsive to chelation |
| Alpha-1 antitrypsin deficiency (AATD) | SERPINA1 (AAT protein) | Cirrhosis or emphysema (liver transplant alone; lung transplant may also be needed) |
| Hereditary hemochromatosis | HFE (iron regulator) | Cirrhosis or HCC (transplant outcomes historically worse due to cardiac iron deposition) |
| Tyrosinemia type 1 | FAH (enzyme in tyrosine metabolism) | If unresponsive to nitisinone (NTBC) |
| Glycogen storage diseases | Various | Severe metabolic instability or liver tumors |
| Crigler-Najjar syndrome type 1 | UGT1A1 (bilirubin conjugation) | Life-threatening bilirubin neurotoxicity (transplant normalizes bilirubin) |
| Familial amyloidotic polyneuropathy (FAP) | TTR (transthyretin) | To stop production of mutant amyloid protein (domino transplant: recipient receives a new liver; recipient’s explanted liver may be transplanted into an older patient with HCC) |
Part 3: When Is Transplant Not an Option? – Contraindications
Just as important as knowing when transplant is indicated is knowing when it is not appropriate. Absolute and relative contraindications vary by transplant center, but general guidelines include:
Absolute Contraindications (Most Centers)
| Contraindication | Rationale |
|---|---|
| Active, untreated sepsis | Immunosuppression after transplant would allow infection to spread uncontrollably |
| Metastatic cancer outside the liver | Transplant does not cure disseminated cancer; immunosuppression may accelerate growth |
| Advanced cardiopulmonary disease | Patient cannot survive the surgery or tolerate immunosuppression (e.g., severe pulmonary hypertension with mean PAP > 50 mmHg) |
| Active substance use disorder (alcohol, opioids, cocaine) without rehabilitation commitment | High risk of relapse, non-adherence to medications, and graft loss |
| Severe, irreversible brain injury | Patient lacks capacity for informed consent and post-transplant self-care |
| HIV/AIDS with low CD4 count (< 100 cells/μL) or uncontrolled opportunistic infections | Historically an absolute contraindication; now relative with effective antiretroviral therapy |
| Multi-organ failure not amenable to multi-organ transplant | Liver transplant alone will not save the patient |
Relative Contraindications (Center-Dependent)
| Contraindication | Nuance |
|---|---|
| Advanced age | Chronological age alone is not a contraindication; biologic age (frailty, comorbidities) matters more. Many centers transplant selected patients up to age 75-80. |
| Obesity (BMI > 40) | Increased surgical risk; may require weight loss before listing |
| Poorly controlled diabetes | Increases infection risk; requires optimization |
| Chronic kidney disease | May require combined liver-kidney transplant if eGFR < 30-40 mL/min |
| HIV infection with controlled viral load | Now a relative indication; outcomes similar to HIV-negative patients |
| Previous malignancy with high recurrence risk | Standard waiting period (e.g., 2-5 years cancer-free) required |
| Non-adherence to medical care | Predicts post-transplant non-adherence to immunosuppression |
| Limited social support | Requires caregiver for post-transplant recovery |
Part 4: How Patients Are Prioritized – The MELD Score
In the United States, patients with chronic liver disease are prioritized for transplant using the Model for End-Stage Liver Disease (MELD) score. The MELD score ranges from 6 (lowest urgency) to 40 (highest urgency) and is calculated using three routine blood tests:
- Creatinine (measures kidney function)
- Bilirubin (measures liver’s ability to excrete bile)
- INR (measures blood clotting)
The formula is:
MELD = 3.78 × ln(creatinine) + 11.2 × ln(INR) + 9.57 × ln(bilirubin) + 6.43
For patients on dialysis, creatinine is automatically set to 4.0 mg/dL. A separate MELD-Na score incorporates serum sodium, which adds prognostic value in patients with ascites and hyponatremia.
MELD Score Interpretation
| MELD Score | 3-Month Mortality Risk | Typical Clinical Status |
|---|---|---|
| < 10 | < 5% | Compensated cirrhosis; not yet transplant-eligible |
| 10-14 | 5-15% | Early decompensation; may be listed but low priority |
| 15-19 | 15-30% | Moderate urgency; often listed for transplant |
| 20-29 | 30-50% | High urgency; hospitalized patients |
| 30-39 | 50-70% | Very high urgency; ICU patients |
| ≥ 40 | > 70% | Status 1 equivalent; urgent transplant needed |
MELD Exceptions
Some liver diseases cause severe symptoms but are not accurately reflected by the MELD score. These patients may receive MELD exception points to increase their priority:
| Disease | MELD Exception |
|---|---|
| Hepatocellular carcinoma (HCC) within Milan Criteria | Automatic MELD 28 (or center-specific equivalent) after 6 months waiting time |
| Hepatic artery thrombosis (post-transplant) | Status 1 or MELD exception |
| Primary hyperoxaluria | MELD exception |
| Cystic fibrosis liver disease | MELD exception |
| Hepatopulmonary syndrome (low oxygen due to liver disease) | MELD exception |
| Portopulmonary hypertension (controlled with therapy) | MELD exception |
| Familial amyloidotic polyneuropathy | MELD exception |
MELD 3.0
In 2023, the Organ Procurement and Transplantation Network (OPTN) implemented MELD 3.0, which adds sex adjustment (females have lower serum creatinine for the same kidney function, disadvantaging them under MELD-Na). MELD 3.0 also incorporates albumin and refines the creatinine calculation. This change has reduced sex-based disparity in liver transplant access.
Part 5: Acute Liver Failure – A Different Prioritization System
Patients with acute liver failure (ALF) are not prioritized by MELD score because their disease progresses too rapidly. Instead, they are designated Status 1 —the highest priority, allowing them to receive the next available liver from a deceased donor within their blood type.
Status 1 criteria (acute liver failure):
- Onset of hepatic encephalopathy within 8 weeks of first symptoms of liver disease
- Absence of pre-existing liver disease
- Life expectancy without transplant < 7 days
- Meeting at least one of:
- INR > 6.5
- Ventilator dependence
- Renal replacement therapy (dialysis)
- Severe acidosis (pH < 7.25 or lactate > 4 mmol/L)
Status 1A (highest tier) is reserved for patients with acute liver failure who are in the ICU and expected to die within hours to days.
Part 6: Living Donor Liver Transplantation – An Alternative to Waiting
Most liver transplants in the United States use deceased donor livers—organs from someone who has died, typically a victim of brain death (DBD) or circulatory death (DCD). However, because the liver regenerates, living donor liver transplantation (LDLT) is possible: a healthy person donates a portion (usually the right lobe, about 60% of the liver) to a recipient.
Advantages of LDLT
- Shorter waiting time (weeks to months instead of years)
- Elective scheduling (surgery occurs when both donor and recipient are optimally prepared)
- Better organ quality (living donor livers have minimal cold ischemia time and come from healthy individuals)
- Lower risk of hepatitis C transmission (donor is screened negative)
- Potential for pre-emptive transplant before decompensation
Disadvantages and Risks
- Donor risk: Major hepatectomy carries a 0.1-0.5% mortality risk for the donor (1 in 200 to 1 in 1000) and 15-30% risk of minor complications (bile leak, wound infection, incisional hernia)
- Recipient risk: Smaller graft (60% of a normal liver) may be insufficient for a large recipient; risk of “small-for-size syndrome”
- Psychosocial complexity: Donor must be fully informed, psychologically stable, and free from coercion
Who Is a Candidate for LDLT?
- Recipients with MELD 15-25 (sick enough to need transplant but not sick enough to get a deceased donor quickly)
- Recipients with HCC who cannot wait 6 months for MELD exception points
- Recipients with hepatopulmonary syndrome or portopulmonary hypertension (high waitlist mortality)
- Small children (pediatric recipients) for whom deceased donor livers are scarce
LDLT is particularly common in countries where deceased donation is rare (e.g., Japan, Korea, India, Turkey). In the United States, LDLT accounts for approximately 5% of adult liver transplants.
Part 7: Outcomes After Liver Transplant
Liver transplantation is one of the most successful solid organ transplants, with excellent short- and long-term survival.
Survival Rates (United States, OPTN Data 2024)
| Time Point | Graft Survival | Patient Survival |
|---|---|---|
| 3 months | 92% | 94% |
| 1 year | 88% | 91% |
| 3 years | 80% | 84% |
| 5 years | 73% | 78% |
| 10 years | 60% | 65% |
Best Outcomes By Indication
| Indication | 5-Year Survival |
|---|---|
| Primary biliary cholangitis (PBC) | 85-90% |
| Primary sclerosing cholangitis (PSC) | 80-85% |
| Alcohol-related liver disease (ALD) | 80-85% |
| Hepatitis C (DAA era) | 80-85% |
| Hepatocellular carcinoma (Milan Criteria) | 70-80% |
| MASLD/MASH | 70-75% |
| Acute liver failure | 80-90% (if transplanted) |
| Retransplantation | 50-60% |
Causes of Graft Loss
| Early (< 1 year) | Late (> 1 year) |
|---|---|
| Primary non-function (PNF) – graft never works | Chronic rejection |
| Hepatic artery thrombosis (HAT) | Disease recurrence (HCV, PBC, PSC, NASH, alcohol) |
| Bile duct complications (leak, stricture) | De novo malignancies (post-transplant lymphoproliferative disease, skin cancer) |
| Acute cellular rejection | Cardiovascular disease (accelerated by immunosuppression) |
| Infection (bacterial, fungal, viral) | Renal failure from calcineurin inhibitor toxicity |
Part 8: Life After Transplant – Immunosuppression and Long-Term Management
A liver transplant is not a cure; it is a treatment exchange. The patient trades end-stage liver disease for a lifetime of immunosuppression and its associated risks.
Standard Immunosuppression Regimen (Triple Therapy)
| Drug Class | Example | Mechanism | Key Side Effects |
|---|---|---|---|
| Calcineurin inhibitor (CNI) | Tacrolimus (Prograf) | Inhibits T-cell activation | Nephrotoxicity, neurotoxicity, hypertension, diabetes, tremor |
| Antimetabolite | Mycophenolate mofetil (CellCept) | Inhibits lymphocyte proliferation | Diarrhea, leukopenia, teratogenicity |
| Corticosteroid | Prednisone | Broad anti-inflammatory | Weight gain, osteoporosis, hyperglycemia, insomnia |
Tapering: Most centers wean prednisone off by 3-6 months post-transplant. Tacrolimus is continued lifelong, often as monotherapy or with mycophenolate.
Long-Term Risks
| Complication | Incidence | Management |
|---|---|---|
| Chronic kidney disease | 30-50% at 5-10 years | Minimize CNI exposure; switch to belatacept or everolimus; consider kidney transplant |
| Post-transplant diabetes mellitus (PTDM) | 20-30% | Lifestyle modification; oral hypoglycemics; insulin |
| Hypertension | 50-70% | Antihypertensives (CCB, ACEi, ARB); reduce CNI dose |
| Hyperlipidemia | 40-60% | Statins; diet; exercise |
| De novo malignancy | 10-15% at 10 years | Skin cancer surveillance (annual dermatology); screening for lymphoma, lung, colon, breast cancer |
| Osteoporosis | 20-40% | Calcium + vitamin D; bisphosphonates if indicated |
| Infections | Common in first year | Prophylaxis (valganciclovir for CMV; TMP-SMX for PJP); vaccinations |
Part 9: The Future – Alternatives to Transplant
While transplant remains the only definitive treatment for end-stage liver disease, several emerging therapies may reduce the need for transplantation or extend the viability of donor organs.
1. Artificial Liver Support Systems
Devices such as the Molecular Adsorbent Recirculating System (MARS) and Prometheus can temporarily remove albumin-bound toxins (bilirubin, bile acids, ammonia) from the blood. They are used as “bridges to transplant” in acute liver failure or acute-on-chronic liver failure (ACLF), buying time for native liver recovery or until a donor organ becomes available. No device yet replaces all liver functions or obviates transplant.
2. Xenotransplantation
In 2022, the first genetically modified pig-to-human liver xenotransplant was performed on a brain-dead recipient. Modifications include:
- Knockout of the alpha-gal gene (prevents hyperacute rejection)
- Insertion of human complement regulatory proteins (CD55, CD59)
- Insertion of human thrombomodulin (prevents coagulation dysregulation)
Clinical trials of pig liver xenotransplantation in living humans are likely within 5-10 years. If successful, xenotransplantation could eliminate the organ shortage.
3. Hepatocyte Transplantation and Liver Cell Therapy
Infusion of isolated human hepatocytes into the spleen or liver can temporarily correct metabolic diseases (e.g., Crigler-Najjar, urea cycle defects). However, hepatocytes do not engraft permanently, and the effect wears off. Cell encapsulation technologies and induced pluripotent stem cell (iPSC)-derived hepatocytes are under investigation.
4. Normothermic Machine Perfusion
Traditionally, donor livers are stored on ice (cold ischemia), which damages the organ. Normothermic machine perfusion (NMP) pumps warm, oxygenated, nutrient-rich blood through the donor liver, preserving it in a near-physiologic state. NMP allows:
- Assessment of liver function before transplant (reducing discard rates)
- Repair of damaged livers (e.g., reducing steatosis)
- Extended preservation time (from 12 to 24+ hours)
- Potential for ex vivo gene therapy or defatting
NMP is increasingly used in high-volume transplant centers.
Summary Table: When Is a Liver Transplant the Only Option?
| Clinical Scenario | Transplant Indicated? | Notes |
|---|---|---|
| Compensated cirrhosis (MELD < 15, no ascites, no encephalopathy) | No | Manage underlying cause; monitor for progression |
| Decompensated cirrhosis (ascites, variceal bleed, encephalopathy, jaundice) | Yes, evaluation required | Transplant is the only definitive treatment; 1-2 year median survival without transplant |
| Acute liver failure (encephalopathy within 8 weeks) | Yes, urgent | Status 1 listing; 80% mortality without transplant |
| Hepatocellular carcinoma within Milan Criteria | Yes | Transplant offers 70-80% 5-year survival |
| Hepatocellular carcinoma outside Milan Criteria | Possibly, after downstaging | Direct transplant has high recurrence risk (> 50%) |
| Primary sclerosing cholangitis with cirrhosis | Yes | No effective medical therapy; high risk of cholangiocarcinoma |
| Primary biliary cholangitis failing UDCA | Yes | Transplant is definitive |
| Alcohol-related cirrhosis with 6 months abstinence | Yes | Excellent outcomes; requires rehabilitation commitment |
| Active, severe alcoholic hepatitis (Lille score > 0.45) | Selected patients | “Early transplant” controversial but increasingly offered |
| MASLD cirrhosis with obesity and diabetes | Yes, after optimization | Weight loss and diabetes control before listing improve outcomes |
| Wilson’s disease with acute liver failure | Yes, urgent | Chelation therapy ineffective in ALF |
| Metabolic diseases (tyrosinemia, Crigler-Najjar, hyperoxaluria) | Yes | Transplant corrects metabolic defect |
| Hepatopulmonary syndrome (PaO2 < 60 mmHg) | Yes | MELD exception; transplant cures hypoxemia |
| Portopulmonary hypertension (mPAP > 35 mmHg) | Possibly, after medical control | Uncontrolled POPH is a contraindication |
| Active substance use without rehabilitation | No | Must complete rehab and demonstrate abstinence |
| Metastatic cancer outside liver | No | Transplant does not cure disseminated cancer |
| Advanced cardiac or pulmonary disease | No (or combined transplant) | High perioperative mortality |
Conclusion: The Last Best Option
Liver transplantation is a remarkable achievement of modern medicine—a procedure that takes a failing, scarred organ from a dying patient and replaces it with a healthy one from a donor, often restoring near-normal quality of life for decades. But it is also a scarce, expensive, and risky intervention. The decision to list a patient for transplant is never taken lightly.
For patients with compensated cirrhosis, the focus is on preventing progression: treating the underlying liver disease, screening for varices and HCC, and avoiding hepatotoxins (alcohol, acetaminophen, certain herbs). For those with decompensated cirrhosis or acute liver failure, the conversation shifts to transplant evaluation, MELD score optimization, and preparing for the possibility of living or deceased donor transplantation.
The good news is that the field is advancing rapidly. New antiviral cures (hepatitis C), better management of MASLD, and emerging technologies like normothermic perfusion and xenotransplantation may one day make liver failure a preventable or reversible condition. Until then, for the patient with end-stage liver disease who has failed all medical therapy, a liver transplant remains the only option—and a life-saving one.
Disclaimer: This article is for informational and educational purposes only and does not constitute medical advice. Liver transplant candidacy decisions are highly individualized and should be made in consultation with a hepatologist and transplant surgeon