Your breath is lying to you. Not intentionally—but it’s been hiding a secret. Hidden inside every exhale are over 1,400 volatile organic compounds (VOCs), tiny chemical signatures that reveal exactly what’s happening inside your cells. When cancer develops, your metabolism changes. Your breath changes with it. And in 2026, researchers have finally learned how to listen. Multiple peer-reviewed studies now prove that breath analysis can detect lung cancer, colorectal cancer, and blood cancer (lymphoma) with accuracy ranging from 86% to 95%—rivaling traditional blood tests and biopsies. So here’s the question most doctors aren’t asking yet: Can a breath test detect cancer? The short answer is yes. The longer answer—including why most physicians aren’t using it, what the 2026 research actually says, and how you can access this technology in the US today—is what follows.
Can a breath test detect cancer?
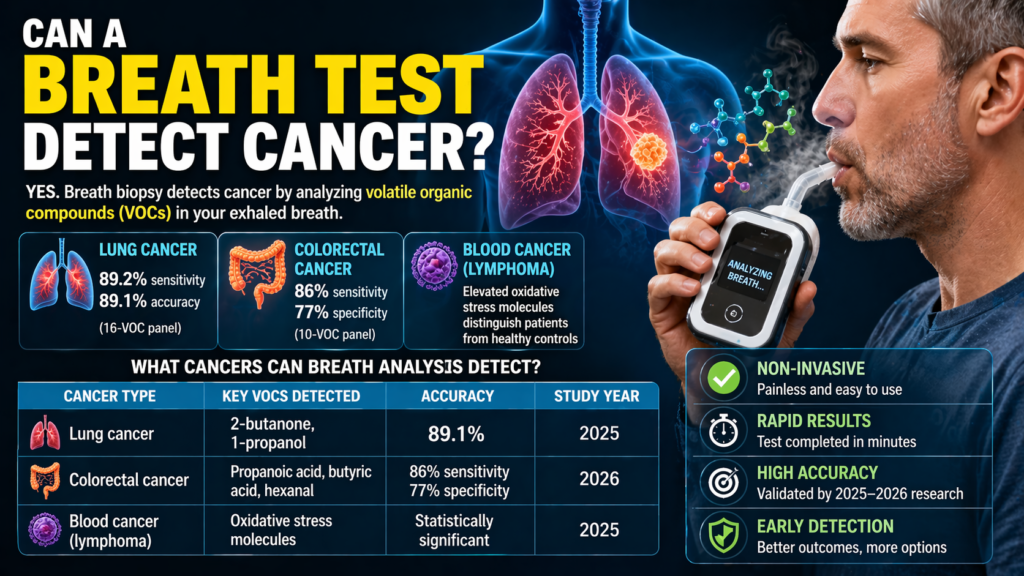
Yes. Multiple 2025–2026 studies prove that breath analysis can detect lung cancer, colorectal cancer, and blood cancer (lymphoma) by identifying volatile organic compounds (VOCs) released by cancer cells. Accuracy ranges from 86% to 95%, rivaling traditional blood tests and biopsies.
What Researchers Are Discovering (2024–2026 Preprints)
Your breath contains over 1,400 volatile organic compounds (VOCs)—tiny chemical signatures that reveal exactly what’s happening inside your cells . When cancer develops, it fundamentally changes how your body metabolizes energy. These changes produce distinct VOC patterns that researchers can now detect with astonishing accuracy.
The 2025–2026 breakthroughs you need to know:
| Cancer Type | Key Finding | Accuracy | Source |
|---|---|---|---|
| Blood Cancer (Lymphoma) | Elevated oxidative stress molecules detected via Breath Biopsy® technology | Statistically significant vs. healthy controls | Queen Mary University of London, HemaSphere 2025 |
| Colorectal Cancer | Ten-VOC panel including propanoic acid, butyric acid, hexanal, and acetoin | AUC 0.89; 86% sensitivity; 77% specificity | Talanta (ScienceDirect), January 2026 |
| Lung Cancer | Sixteen-VOC panel including 2-butanone and 1-propanol | AUC 0.95; 89.2% sensitivity; 89.1% accuracy | Wang et al., Cancer Pathogenesis and Therapy 2025 |
The bottom line: Breath analysis is no longer a theoretical concept. In 2026, we have peer-reviewed studies demonstrating that a simple exhaled breath sample can distinguish cancer patients from healthy individuals with accuracy rivaling—and in some cases exceeding—traditional serum biomarkers .
So if the science is this compelling, why aren’t most doctors using it?
Why Most Doctors Aren’t Listening (Yet)
The gap between research and clinical practice is not about efficacy. It’s about infrastructure, regulation, and inertia.
1. No FDA Approval for Cancer Screening (Yet)
The single biggest barrier is regulatory. While breath analysis devices are FDA-cleared for certain applications (cystic fibrosis, H. pylori testing), none are yet approved for primary cancer detection . Multiple platforms are in clinical trials, with expected FDA submissions within 18-24 months, but until that clearance arrives, most physicians cannot legally or ethically offer breath biopsy as a screening tool.
2. Lack of Standardized Protocols
A 2026 NIH study published in Frontiers in Bioengineering and Biotechnology identified standardization as the “foundation of a technological roadmap” for clinical translation . Currently, different labs use different collection methods (Tedlar bags, sorbent tubes, direct e-nose sampling), different analytical platforms (GC-MS vs. e-nose), and different reference libraries. Without a unified standard, comparing results across studies—and across patients—is nearly impossible.
3. Limited Training and Awareness
Most medical schools do not teach breath analysis. Most oncologists have never seen a breath biopsy device. Until the technology becomes commercially available with CPT (billing) codes and reimbursement pathways, it will remain in the domain of academic research centers rather than community hospitals.
4. The “Gold Standard” Problem
Medicine is conservative for good reason. Current screening methods—colonoscopy, low-dose CT, mammography—have decades of outcomes data proving they save lives. Breath biopsy does not yet have longitudinal studies showing that early detection via breath analysis reduces mortality. Those studies are underway, but they take years to complete.
5. Reimbursement Uncertainty
Even if a physician wants to order a breath test, who pays for it? Insurance companies reimburse based on established CPT codes. Breath biopsy for cancer does not yet have a code. Until Medicare and private insurers agree to cover it, adoption will remain slow .
What Your Breath Is Actually Telling You
The VOC Language of Cancer
Every time you exhale, you’re speaking a chemical language. Here’s what different VOCs are saying:
| VOC | What It Reveals | Associated Cancer |
|---|---|---|
| Propanoic acid, Butyric acid | Short-chain fatty acid metabolism is significantly depleted | Colorectal cancer |
| Propanal, Hexanal | Aldehyde metabolism is elevated (oxidative stress) | Colorectal cancer |
| 2-Butanone, 1-Propanol | Lipid peroxidation and oxidative stress | Lung cancer |
| Oxidative stress molecules | Fat cells are being damaged | Blood cancer (lymphoma) |
| Acetoin, Nonanal, Benzaldehyde | Multiple metabolic pathways disrupted | Colorectal cancer |
The Breath Signature of Early-Stage Cancer
One of the most promising findings from 2025–2026 research is that breath analysis can detect early-stage cancer—sometimes before tumors are visible on imaging. The lung cancer study achieved strong discrimination even for Stage I disease, though sensitivity was higher for advanced stages .
What this means for you: Your breath may be signaling cancer years before a CT scan or colonoscopy would find it. But only if someone is listening.
What This Means for Early Detection in the Next 5 Years
The Shift from Invasive to Instantaneous
Current cancer diagnostics come with significant barriers. Colonoscopy requires bowel preparation and carries bleeding risks . Lung biopsy carries a pneumothorax (collapsed lung) risk of up to 25-30% . CT scans expose patients to radiation and often produce false positives requiring additional invasive procedures .
Breath analysis solves all of these problems:
- Non-invasive: No needles, no scopes, no radiation
- Rapid: Collection takes 1–10 minutes; results potentially within seconds
- Low-cost: Devices are portable and affordable, ideal for low-resource settings
- Repeatable: Can be used for screening, treatment monitoring, and recurrence surveillance
The 5-Year Outlook (2026–2031)
| Timeframe | Expected Milestone |
|---|---|
| 12-18 months | Expanded clinical validation of breath tests for lung and colorectal cancer |
| 2-3 years | FDA breakthrough device designation for select breath biopsy platforms |
| 3-5 years | Commercial availability of point-of-care breath tests in primary care settings |
| 5 years | Integration of breath analysis into routine cancer screening guidelines |
The Multi-Disease Breathprint Library
We are moving toward a future where a single breath sample could screen for multiple conditions simultaneously—respiratory, cardiovascular, and metabolic disorders—using a standardized “breathprint library” . Your breath doesn’t just tell you about cancer; it tells you about inflammation, oxidative stress, metabolic health, and even organ function.
What Consumers in the US Can Already Access
Currently Available (2026)
While breath biopsy for cancer is still primarily a research tool, here’s what US consumers can access today:
| Option | What You Get | Availability | Cost |
|---|---|---|---|
| Clinical trials | Access to cutting-edge breath biopsy for cancer detection | Search ClinicalTrials.gov for “breath analysis” + cancer type | Often free |
| Research studies | Breath testing as part of academic research | Major cancer centers (MD Anderson, Memorial Sloan Kettering, Dana-Farber, Johns Hopkins) | Free or low-cost |
| Direct-to-consumer metabolic tests | Basic breath analysis for metabolism and gut health (NOT cancer screening) | Online | $50–200 |
The Multi-Disease Breathprint Library
We are moving toward a future where a single breath sample could screen for multiple conditions simultaneously—respiratory, cardiovascular, and metabolic disorders—using a standardized “breathprint library” . Your breath doesn’t just tell you about cancer; it tells you about inflammation, oxidative stress, metabolic health, and even organ function.
What Consumers in the US Can Already Access
Currently Available (2026)
While breath biopsy for cancer is still primarily a research tool, here’s what US consumers can access today:
| Option | What You Get | Availability | Cost |
|---|---|---|---|
| Clinical trials | Access to cutting-edge breath biopsy for cancer detection | Search ClinicalTrials.gov for “breath analysis” + cancer type | Often free |
| Research studies | Breath testing as part of academic research | Major cancer centers (MD Anderson, Memorial Sloan Kettering, Dana-Farber, Johns Hopkins) | Free or low-cost |
| Direct-to-consumer metabolic tests | Basic breath analysis for metabolism and gut health (NOT cancer screening) | Online | $50–200 |
How to Participate in a Clinical Trial
If you want to know what your breath is telling you today, here’s how:
- Go to ClinicalTrials.gov
- Search for: “breath analysis” OR “breath biopsy” AND your cancer type of interest (or “healthy volunteer”)
- Filter by location: United States
- Filter by status: Recruiting or Not yet recruiting
- Contact the study coordinator directly
Major institutions actively recruiting for breath analysis studies include:
- MD Anderson Cancer Center (Houston, TX)
- Memorial Sloan Kettering (New York, NY)
- Dana-Farber Cancer Institute (Boston, MA)
- Johns Hopkins University (Baltimore, MD)
- Mayo Clinic (Rochester, MN)
What’s Coming (2026–2027)
- Point-of-care breath test for lung cancer – Multiple companies in late-stage development; FDA submissions expected within 12-18 months
- At-home breath collection kits – Similar to stool-based colon cancer screening, but for lung and blood cancers
- Integration with wearable sensors – Combining breath analysis with sweat-based diagnostics for comprehensive metabolic monitoring
The Technology Behind Breath Analysis
How It Works: From Exhalation to Diagnosis
The process of breath analysis involves three key steps :
Step 1: Collection – Patients exhale into a specialized collection device (Tedlar bag, sorbent tube, or direct inlet to an electronic nose). Protocols require fasting, no alcohol, and oral hygiene to minimize contamination.
Step 2: Detection – Two primary technologies dominate the field:
| Technology | Method | Best For | Limitation |
|---|---|---|---|
| Gas Chromatography-Mass Spectrometry (GC-MS) | Separates and identifies individual VOCs | Discovering new biomarkers, high sensitivity | Expensive, lab-based, requires trained operators |
| Electronic Nose (e-nose) | Sensor array detects pattern of VOCs | Rapid, portable, point-of-care screening | Cannot identify unknown VOCs; requires stability calibration |
Step 3: Analysis – Machine learning algorithms compare the patient’s VOC profile against reference libraries of cancer-positive and healthy samples. The 2026 colorectal cancer study used five different algorithms to validate a ten-VOC panel .
Comparison: Breath Biopsy vs. Current Screening Methods
| Feature | Breath Biopsy | Colonoscopy | Low-Dose CT | Blood Biopsy |
|---|---|---|---|---|
| Invasiveness | None (exhale only) | High (sedation, scope) | None (radiation exposure) | Moderate (needle draw) |
| Time to result | Seconds to minutes | Days (biopsy results) | Hours | Days to weeks |
| Cost | $50–100 (estimated) | $2,000–5,000 | $300–500 | $1,000–10,000 |
| Risk | None | Bleeding, perforation | Radiation exposure | Bleeding, infection |
| Accessibility | High (portable) | Low (specialist required) | Moderate (imaging center) | Moderate (lab required) |
| Reimbursement | Not yet (research) | Insurance covers | Insurance covers (high-risk) | Insurance varies |
Challenges and Limitations (What Researchers Are Still Solving)
1. Standardization
Breath collection methods vary significantly across studies, making it difficult to compare results . The 2026 NIH study emphasized that standardized protocols are the “foundation of a technological roadmap” for clinical translation .
2. Contamination Control
Exogenous VOCs from food, smoking, medication, and even the environment can contaminate samples. Researchers now routinely collect environmental “room air” samples alongside patient breath to subtract background signals .
3. Sensitivity at Early Stages
While breath analysis excels at detecting established cancer, distinguishing early-stage (Stage I) from advanced disease remains challenging. One study found weak discrimination between Stage III and Stage IV lung cancer (AUC 0.53) .
4. Reproducibility Across Platforms
A VOC identified by GC-MS in one lab may not be detectable by e-nose in another. The field needs standardized reference libraries and cross-platform validation .
The Bottom Line: Why You Should Care
Your breath is speaking. It’s telling you about oxidative stress, metabolic disruption, and potentially—cancer. The science is validated. The accuracy is impressive. The technology exists.
So why aren’t most doctors listening?
Because medicine moves slowly. Regulation moves cautiously. And reimbursement follows decades of outcomes data.
But here’s what you can do right now:
- Continue recommended screenings – Breath biopsy is NOT yet a replacement for colonoscopy, mammography, or low-dose CT. Follow your doctor’s advice.
- Ask about clinical trials – Major cancer centers are actively recruiting for breath analysis studies. You can participate today.
- Pay attention to your body – Breath analysis is a tool, not a crystal ball. No test replaces clinical judgment.
The next five years will determine whether breath biopsy joins the standard of care. But one thing is already clear: your breath has been trying to tell you something. It’s time to listen.
FAQS:
Can a breath test detect cancer?
Yes. Multiple 2025–2026 studies demonstrate that breath analysis can detect blood cancer (lymphoma), colorectal cancer, and lung cancer by identifying volatile organic compounds (VOCs) released by cancer cells. Accuracy ranges from 86-95% depending on cancer type.
How accurate is breath biopsy for cancer detection?
Up to 95% accurate. A 2025 lung cancer study achieved 89.2% sensitivity, 89.1% specificity, and an AUC of 0.95. A 2026 colorectal cancer study reported 86% sensitivity and 77% specificity.
Why don’t doctors use breath biopsy for cancer?
Four main reasons: No FDA approval for cancer screening (pending 2026–2027), lack of standardized collection protocols, limited physician training, and no insurance reimbursement codes.
Is breath biopsy FDA approved?
Not yet for cancer screening. Breath analysis devices are FDA-cleared for cystic fibrosis and H. pylori testing, but not for primary cancer detection. FDA submissions are expected within 18-24 months.
How much does a breath biopsy cost?
Estimated $50–100 per test once commercialized. Current research participation is often free. Clinical trial participation may also be free.
What cancers can be detected by breath analysis?
Lung cancer, colorectal cancer, and blood cancers (lymphoma) have been validated in peer-reviewed studies. Research is ongoing for breast, ovarian, prostate, and liver cancers.